Garcinol Encapsulated Ph-Sensitive Biodegradable Nanoparticles: A Novel Therapeutic Strategy for the Treatment of Inflammatory Bowel Disease
- PMID: 33799680
- PMCID: PMC7999919
- DOI: 10.3390/polym13060862
Garcinol Encapsulated Ph-Sensitive Biodegradable Nanoparticles: A Novel Therapeutic Strategy for the Treatment of Inflammatory Bowel Disease
Abstract
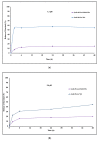
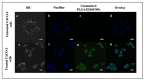
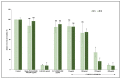
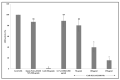
The emergence of pH-sensitive nanoscale particles is beneficial due to their ability to only release cargo in a colonic pH environment, which helps to directly target inflamed tissues in inflammatory bowel disease (IBD). Hence, we have designed the formulation of pH-sensitive biodegradable garcinol (GAR)-loaded poly (lactic-co-glycolic acid) (PLGA) coated with Eudragit® S100 (ES100) (GAR-PLGA-ES100 nanoparticles (NPs)) for reducing inflammation caused by proinflammatory cytokines. The GAR-PLGA-ES100 NPs were prepared using a solvent evaporation technique and characterized for shape and surface morphology. An in vitro drug release study revealed the release of the drug specifically from NPs at the colonic pH of 7.4. The in vitro cytotoxicity of the GAR-PLGA-ES100 NPs was also evaluated and found to be highly biocompatible with CACO-2 cells. These NPs were able to reduce lactate dehydrogenase (LDH) and myeloperoxidase (MPO) activity. Inhibition of the expression of pro-inflammatory cytokine TNF-α , chemokine interleukin (IL)-8 and the nuclear factor kappa light chain enhancer of activated B-cells (NF-κB) was observed after GAR-PLGA-ES100 NPs treatment. Therefore, our results support the idea that GAR-PLGA-ES100 NPs show substantial improvement after the release of the drug, specifically in colonic pH targeting and reduction in the activation of inflammation that leads to IBD, suggesting that GAR-PLGA-ES100 NPs are promising candidates for oral delivery to colonic inflamed tissue.
Keywords: eudragit S100; garcinol; inflammatory bowel disease; nuclear factor kappa light chain enhancer of activated B-cells; pH-sensitive nanoparticles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Oral targeted delivery of Imatinib by pH responsive copolymer modulates liver fibrosis in the mice model.Int J Pharm. 2023 Jun 25;641:123068. doi: 10.1016/j.ijpharm.2023.123068. Epub 2023 May 22. Int J Pharm. 2023. PMID: 37225027
-
pH-Sensitive Nanoparticles for Colonic Delivery Anti-miR-301a in Mouse Models of Inflammatory Bowel Diseases.Nanomaterials (Basel). 2023 Oct 20;13(20):2797. doi: 10.3390/nano13202797. Nanomaterials (Basel). 2023. PMID: 37887947 Free PMC article.
-
Preparation and characterization of pH-sensitive nanoparticles of budesonide for the treatment of ulcerative colitis.Drug Des Devel Ther. 2018 Aug 22;12:2601-2609. doi: 10.2147/DDDT.S170676. eCollection 2018. Drug Des Devel Ther. 2018. PMID: 30174414 Free PMC article.
-
pH-sensitive nanoparticles for colonic delivery of curcumin in inflammatory bowel disease.Int J Pharm. 2014 Oct 1;473(1-2):203-12. doi: 10.1016/j.ijpharm.2014.07.009. Epub 2014 Jul 8. Int J Pharm. 2014. PMID: 25014369
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
Cited by
-
Recent Advances in Garcinia cambogia Nutraceuticals in Relation to Its Hydroxy Citric Acid Level. A Comprehensive Review of Its Bioactive Production, Formulation, and Analysis with Future Perspectives.ACS Omega. 2022 Jul 19;7(30):25948-25957. doi: 10.1021/acsomega.2c02838. eCollection 2022 Aug 2. ACS Omega. 2022. PMID: 35936438 Free PMC article. Review.
-
Garcinol-Attenuated Gastric Ulcer (GU) Experimentally Induced in Rats Via Affecting Inflammation, Cell Proliferation, and DNA Polymerization.Cureus. 2023 Aug 11;15(8):e43317. doi: 10.7759/cureus.43317. eCollection 2023 Aug. Cureus. 2023. PMID: 37577271 Free PMC article.
-
Nanoparticle-Based Drug Delivery Systems for Inflammatory Bowel Disease Treatment.Drug Des Devel Ther. 2024 Jul 15;18:2921-2949. doi: 10.2147/DDDT.S461977. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 39055164 Free PMC article. Review.
-
Dual pH and microbial-sensitive galactosylated polymeric nanocargoes for multi-level targeting to combat ulcerative colitis.Asian J Pharm Sci. 2023 Jul;18(4):100831. doi: 10.1016/j.ajps.2023.100831. Epub 2023 Jul 26. Asian J Pharm Sci. 2023. PMID: 37588990 Free PMC article.
-
Ileo-Colon Targeting of the Poorly Water-Soluble Drug Celecoxib Using a pH-Dependent Coating in Combination with Self-Emulsifying Drug Delivery or Solid Dispersion Systems.Pharmaceutics. 2021 May 15;13(5):731. doi: 10.3390/pharmaceutics13050731. Pharmaceutics. 2021. PMID: 34063521 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

