Characterization and Separation of Live and Dead Yeast Cells Using CMOS-Based DEP Microfluidics
- PMID: 33800809
- PMCID: PMC8001765
- DOI: 10.3390/mi12030270
Characterization and Separation of Live and Dead Yeast Cells Using CMOS-Based DEP Microfluidics
Abstract
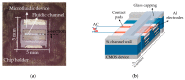
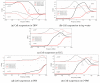
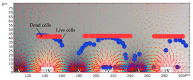
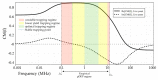
This study aims at developing a miniaturized CMOS integrated silicon-based microfluidic system, compatible with a standard CMOS process, to enable the characterization, and separation of live and dead yeast cells (as model bio-particle organisms) in a cell mixture using the DEP technique. DEP offers excellent benefits in terms of cost, operational power, and especially easy electrode integration with the CMOS architecture, and requiring label-free sample preparation. This can increase the likeliness of using DEP in practical settings. In this work the DEP force was generated using an interdigitated electrode arrays (IDEs) placed on the bottom of a CMOS-based silicon microfluidic channel. This system was primarily used for the immobilization of yeast cells using DEP. This study validated the system for cell separation applications based on the distinct responses of live and dead cells and their surrounding media. The findings confirmed the device's capability for efficient, rapid and selective cell separation. The viability of this CMOS embedded microfluidic for dielectrophoretic cell manipulation applications and compatibility of the dielectrophoretic structure with CMOS production line and electronics, enabling its future commercially mass production.
Keywords: CMOS-based lab-on-a-chip; cell characterization; cell separation; dielectrophoresis (DEP); interdigitated electrodes (IDEs); microfluidics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





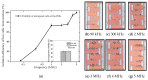

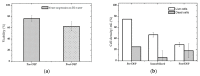
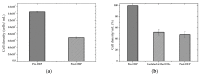
References
-
- Demircan Y., Yilmaz G., Külah H., Demirci U., Khademhosseini A., Langer R., Blander J. Microfluidic Technologies for Human Health. World Scientific; Singapore: 2013. Electrophoresis and Dielectrophoresis for Lab-on-a-Chip (LOC) Analyses; pp. 341–375.
-
- Li H., Zheng Y., Akin D., Bashir R. Characterization and modeling of a microfluidic dielectrophoresis filter for biological species. J. Microelectromechanical Syst. 2005;14:103–112. doi: 10.1109/JMEMS.2004.839124. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

