Biofabrication in Congenital Cardiac Surgery: A Plea from the Operating Theatre, Promise from Science
- PMID: 33800971
- PMCID: PMC8004062
- DOI: 10.3390/mi12030332
Biofabrication in Congenital Cardiac Surgery: A Plea from the Operating Theatre, Promise from Science
Abstract
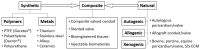
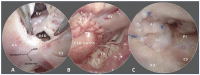
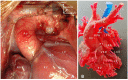
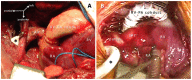
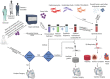
Despite significant advances in numerous fields of biofabrication, clinical application of biomaterials combined with bioactive molecules and/or cells largely remains a promise in an individualized patient settings. Three-dimensional (3D) printing and bioprinting evolved as promising techniques used for tissue-engineering, so that several kinds of tissue can now be printed in layers or as defined structures for replacement and/or reconstruction in regenerative medicine and surgery. Besides technological, practical, ethical and legal challenges to solve, there is also a gap between the research labs and the patients' bedside. Congenital and pediatric cardiac surgery mostly deal with reconstructive patient-scenarios when defects are closed, various segments of the heart are connected, valves are implanted. Currently available biomaterials lack the potential of growth and conduits, valves derange over time surrendering patients to reoperations. Availability of viable, growing biomaterials could cancel reoperations that could entail significant public health benefit and improved quality-of-life. Congenital cardiac surgery is uniquely suited for closing the gap in translational research, rapid application of new techniques, and collaboration between interdisciplinary teams. This article provides a succinct review of the state-of-the art clinical practice and biofabrication strategies used in congenital and pediatric cardiac surgery, and highlights the need and avenues for translational research and collaboration.
Keywords: 3D bioprinting; bioactive molecules; biofabrication; biomaterials; congenital cardiac surgery; congenital heart disease; reconstructive surgery; stem cells; tissue engineering; translational research.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Congenital Heart Disease Statistics 2006, British Heart Foundation. [(accessed on 20 March 2008)]; Available online: www.heartstats.org.
-
- Zimmerman M.S., Smith A.G.C., Sable C.A., Echko M.M., Wilner L.B., Olsen H.E., Atalay H.T., Awasthi A., Bhutta Z.A., Boucher J.L., et al. Global, regional, and national burden of congenital heart disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Child Adolesc. Health. 2020;4:185–200. doi: 10.1016/S2352-4642(19)30402-X. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

