Methamphetamine Blocks Adenosine A2A Receptor Activation via Sigma 1 and Cannabinoid CB1 Receptors
- PMID: 33803075
- PMCID: PMC7963146
- DOI: 10.3390/ijms22052743
Methamphetamine Blocks Adenosine A2A Receptor Activation via Sigma 1 and Cannabinoid CB1 Receptors
Abstract
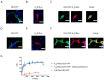
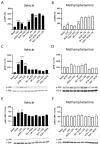
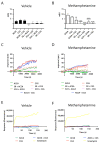
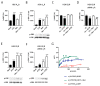
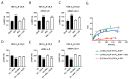
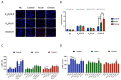
Methamphetamine is, worldwide, one of the most consumed drugs of abuse. One important side effect is neurodegeneration leading to a decrease in life expectancy. The aim of this paper was to check whether the drug affects one of the receptors involved in neurodegeneration/neuroprotection events, namely the adenosine A2A receptor (A2AR). First, we noticed that methamphetamine does not affect A2A functionality if the receptor is expressed in a heterologous system. However, A2AR becomes sensitive to the drug upon complexes formation with the cannabinoid CB1 receptor (CB1R) and the sigma 1 receptor (σ1R). Signaling via both adenosine A2AR and cannabinoid CB1R was affected by methamphetamine in cells co-expressing the two receptors. In striatal primary cultures, the A2AR-CB1R heteromer complex was detected and methamphetamine not only altered its expression but completely blocked the A2AR- and the CB1R-mediated activation of the mitogen activated protein kinase (MAPK) pathway. In conclusion, methamphetamine, with the participation of σ1R, alters the expression and function of two interacting receptors, A2AR, which is a therapeutic target for neuroprotection, and CB1R, which is the most abundant G protein-coupled receptor (GPCR) in the brain.
Keywords: G protein-coupled receptor GPCR; drug of abuse; heteromer; neuroprotection; striatal neurons.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

