Development and Evaluation of 1'-Acetoxychavicol Acetate (ACA)-Loaded Nanostructured Lipid Carriers for Prostate Cancer Therapy
- PMID: 33804975
- PMCID: PMC8063947
- DOI: 10.3390/pharmaceutics13040439
Development and Evaluation of 1'-Acetoxychavicol Acetate (ACA)-Loaded Nanostructured Lipid Carriers for Prostate Cancer Therapy
Abstract
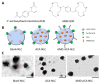
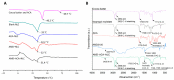
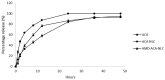
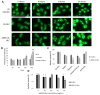
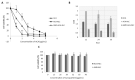
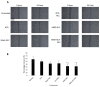
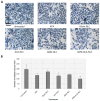
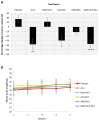
1'-acetoxychavicol acetate (ACA) extracted from the rhizomes of Alpinia conchigera Griff (Zingiberaceae) has been shown to deregulate the NF-ĸB signaling pathway and induce apoptosis-mediated cell death in many cancer types. However, ACA is a hydrophobic ester, with poor solubility in an aqueous medium, limited bioavailability, and nonspecific targeting in vivo. To address these problems, ACA was encapsulated in a nanostructured lipid carrier (NLC) anchored with plerixafor octahydrochloride (AMD3100) to promote targeted delivery towards C-X-C chemokine receptor type 4 (CXCR4)-expressing prostate cancer cells. The NLC was prepared using the melt and high sheer homogenization method, and it exhibited ideal physico-chemical properties, successful encapsulation and modification, and sustained rate of drug release. Furthermore, it demonstrated time-based and improved cellular uptake, and improved cytotoxic and anti-metastatic properties on PC-3 cells in vitro. Additionally, the in vivo animal tumor model revealed significant anti-tumor efficacy and reduction in pro-tumorigenic markers in comparison to the placebo, without affecting the weight and physiological states of the nude mice. Overall, ACA-loaded NLC with AMD3100 surface modification was successfully prepared with evidence of substantial anti-cancer efficacy. These results suggest the potential use of AMD3100-modified NLCs as a targeting carrier for cytotoxic drugs towards CXCR4-expressing cancer cells.
Keywords: 1′-acetoxychavicol acetate; AMD3100; nanostructured lipid carrier; prostate cancer; targeted delivery.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Recombinant human alpha fetoprotein synergistically potentiates the anti-cancer effects of 1'-S-1'-acetoxychavicol acetate when used as a complex against human tumours harbouring AFP-receptors.Oncotarget. 2015 Jun 30;6(18):16151-67. doi: 10.18632/oncotarget.3951. Oncotarget. 2015. PMID: 26158863 Free PMC article.
-
Dual-function nanostructured lipid carriers to deliver IR780 for breast cancer treatment: Anti-metastatic and photothermal anti-tumor therapy.Acta Biomater. 2017 Apr 15;53:399-413. doi: 10.1016/j.actbio.2017.01.070. Epub 2017 Feb 1. Acta Biomater. 2017. PMID: 28159715
-
In vitro inhibitory mechanisms and molecular docking of 1'-S-1'-acetoxychavicol acetate on human cytochrome P450 enzymes.Phytomedicine. 2017 Jul 15;31:1-9. doi: 10.1016/j.phymed.2017.05.002. Epub 2017 May 3. Phytomedicine. 2017. PMID: 28606510
-
Novel purification of 1'S-1'-Acetoxychavicol acetate from Alpinia galanga and its cytotoxic plus antiproliferative activity in colorectal adenocarcinoma cell line SW480.Biomed Pharmacother. 2017 Jul;91:485-493. doi: 10.1016/j.biopha.2017.04.114. Epub 2017 May 4. Biomed Pharmacother. 2017. PMID: 28477464
-
Pharmacological Effects of 1'-Acetoxychavicol Acetate, a Major Constituent in the Rhizomes of Alpinia galanga and Alpinia conchigera.J Med Food. 2020 May;23(5):465-475. doi: 10.1089/jmf.2019.4490. Epub 2020 Feb 18. J Med Food. 2020. PMID: 32069429 Review.
Cited by
-
Recent Advances in Functional Polymer Materials for Energy, Water, and Biomedical Applications: A Review.Polymers (Basel). 2021 Dec 10;13(24):4327. doi: 10.3390/polym13244327. Polymers (Basel). 2021. PMID: 34960878 Free PMC article. Review.
-
Natural Products for Cancer Therapy: A Review of Their Mechanism of Actions and Toxicity in the Past Decade.J Trop Med. 2022 Mar 11;2022:5794350. doi: 10.1155/2022/5794350. eCollection 2022. J Trop Med. 2022. PMID: 35309872 Free PMC article. Review.
References
-
- International Agency for Research on Cancer Global Cancer Observatory [Homepage on the Internet] [(accessed on 30 December 2020)];2020 Available online: https://gco.iarc.fr.
-
- Picus J., Schultz M. Docetaxel (Taxotere) as monotherapy in the treatment of hormone-refractory prostate cancer: Preliminary results. Semin. Oncol. 1999;26:14–18. - PubMed
-
- Kondo A., Ohigashi H., Murakami A., Suratwadee J., Koshimizu K. 1′-acetoxychavicol acetate as a potent inhibitor of tumor promoter-induced Epstein-Barr Virus activation from Languas galanga, a traditional Thai condiment. Biosci. Biotechnol. Biochem. 1993;57:1344–1345. doi: 10.1271/bbb.57.1344. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

