Effectors Targeting the Unfolded Protein Response during Intracellular Bacterial Infection
- PMID: 33805575
- PMCID: PMC8065698
- DOI: 10.3390/microorganisms9040705
Effectors Targeting the Unfolded Protein Response during Intracellular Bacterial Infection
Abstract
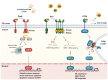
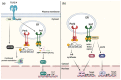
The unfolded protein response (UPR) is a homeostatic response to endoplasmic reticulum (ER) stress within eukaryotic cells. The UPR initiates transcriptional and post-transcriptional programs to resolve ER stress; or, if ER stress is severe or prolonged, initiates apoptosis. ER stress is a common feature of bacterial infection although the role of the UPR in host defense is only beginning to be understood. While the UPR is important for host defense against pore-forming toxins produced by some bacteria, other bacterial effector proteins hijack the UPR through the activity of translocated effector proteins that facilitate intracellular survival and proliferation. UPR-mediated apoptosis can limit bacterial replication but also often contributes to tissue damage and disease. Here, we discuss the dual nature of the UPR during infection and the implications of UPR activation or inhibition for inflammation and immunity as illustrated by different bacterial pathogens.
Keywords: ER stress; UPR; bacteria; effector proteins; infection; secretion systems.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

