Potential of Nutraceutical Supplementation in the Modulation of White and Brown Fat Tissues in Obesity-Associated Disorders: Role of Inflammatory Signalling
- PMID: 33805912
- PMCID: PMC8037903
- DOI: 10.3390/ijms22073351
Potential of Nutraceutical Supplementation in the Modulation of White and Brown Fat Tissues in Obesity-Associated Disorders: Role of Inflammatory Signalling
Abstract
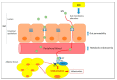
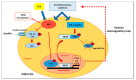
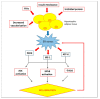
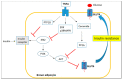
The high incidence of obesity is associated with an increasing risk of several chronic diseases such as cardiovascular disease, type 2 diabetes and non-alcoholic fatty liver disease (NAFLD). Sustained obesity is characterized by a chronic and unsolved inflammation of adipose tissue, which leads to a greater expression of proinflammatory adipokines, excessive lipid storage and adipogenesis. The purpose of this review is to clarify how inflammatory mediators act during adipose tissue dysfunction in the development of insulin resistance and all obesity-associated diseases. In particular, we focused our attention on the role of inflammatory signaling in brown adipose tissue (BAT) thermogenic activity and the browning of white adipose tissue (WAT), which represent a relevant component of adipose alterations during obesity. Furthermore, we reported the most recent evidence in the literature on nutraceutical supplementation in the management of the adipose inflammatory state, and in particular on their potential effect on common inflammatory mediators and pathways, responsible for WAT and BAT dysfunction. Although further research is needed to demonstrate that targeting pro-inflammatory mediators improves adipose tissue dysfunction and activates thermogenesis in BAT and WAT browning during obesity, polyphenols supplementation could represent an innovative therapeutic strategy to prevent progression of obesity and obesity-related metabolic diseases.
Keywords: BAT; WAT; browning; inflammation; metabolic diseases; nutraceutical supplementation; obesity; polyphenols.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Dietary polyphenols and their roles in fat browning.J Nutr Biochem. 2019 Feb;64:1-12. doi: 10.1016/j.jnutbio.2018.09.028. Epub 2018 Oct 13. J Nutr Biochem. 2019. PMID: 30414469 Review.
-
A polyphenolic extract from green tea leaves activates fat browning in high-fat-diet-induced obese mice.J Nutr Biochem. 2017 Nov;49:15-21. doi: 10.1016/j.jnutbio.2017.07.008. Epub 2017 Jul 24. J Nutr Biochem. 2017. PMID: 28863365
-
Melinjo (Gnetum gnemon L.) seed extract induces uncoupling protein 1 expression in brown fat and protects mice against diet-induced obesity, inflammation, and insulin resistance.Nutr Res. 2018 Oct;58:17-25. doi: 10.1016/j.nutres.2018.06.012. Epub 2018 Jul 3. Nutr Res. 2018. PMID: 30340811
-
Many Ways to Rome: Exercise, Cold Exposure and Diet-Do They All Affect BAT Activation and WAT Browning in the Same Manner?Int J Mol Sci. 2022 Apr 26;23(9):4759. doi: 10.3390/ijms23094759. Int J Mol Sci. 2022. PMID: 35563150 Free PMC article. Review.
-
Beiging of white adipose tissue as a therapeutic strategy for weight loss in humans.Horm Mol Biol Clin Investig. 2017 Jun 23;31(2):/j/hmbci.2017.31.issue-2/hmbci-2017-0016/hmbci-2017-0016.xml. doi: 10.1515/hmbci-2017-0016. Horm Mol Biol Clin Investig. 2017. PMID: 28672737 Review.
Cited by
-
Oleanolic Acid-Enriched Olive Oil Alleviates the Interleukin-6 Overproduction Induced by Postprandial Triglyceride-Rich Lipoproteins in THP-1 Macrophages.Nutrients. 2021 Sep 29;13(10):3471. doi: 10.3390/nu13103471. Nutrients. 2021. PMID: 34684472 Free PMC article.
-
Insulin and TLR4 Inhibitor Improve Motor Impairments in a Rat Model of Parkinson's Disease.Iran J Pharm Res. 2024 Jun 11;23(1):e144200. doi: 10.5812/ijpr-144200. eCollection 2024 Jan-Dec. Iran J Pharm Res. 2024. PMID: 39830652 Free PMC article.
-
Nocellara Del Belice (Olea europaea L. Cultivar): Leaf Extract Concentrated in Phenolic Compounds and Its Anti-Inflammatory and Radical Scavenging Activity.Plants (Basel). 2022 Dec 21;12(1):27. doi: 10.3390/plants12010027. Plants (Basel). 2022. PMID: 36616158 Free PMC article.
-
Effects of Bergamot Polyphenols on Mitochondrial Dysfunction and Sarcoplasmic Reticulum Stress in Diabetic Cardiomyopathy.Nutrients. 2021 Jul 20;13(7):2476. doi: 10.3390/nu13072476. Nutrients. 2021. PMID: 34371986 Free PMC article. Review.
-
Browning of Adipocytes: A Potential Therapeutic Approach to Obesity.Nutrients. 2023 May 8;15(9):2229. doi: 10.3390/nu15092229. Nutrients. 2023. PMID: 37432449 Free PMC article. Review.
References
-
- Engin A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Adv. Exp. Med. Biol. 2017;960:1–17. - PubMed
-
- NCD Risk Factor Collaboration (NCD-RisC) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 2017;390:2627–2642. doi: 10.1016/S0140-6736(17)32129-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

