D-α-Tocopherol-Based Micelles for Successful Encapsulation of Retinoic Acid
- PMID: 33806321
- PMCID: PMC7999664
- DOI: 10.3390/ph14030212
D-α-Tocopherol-Based Micelles for Successful Encapsulation of Retinoic Acid
Abstract
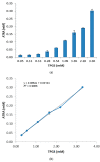
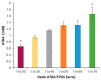
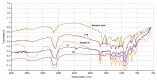
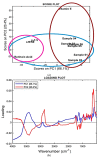
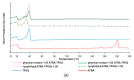
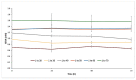
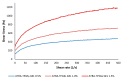
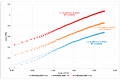
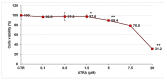
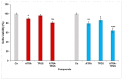
All-trans-retinoic acid (ATRA) represents the first-choice treatment for several skin diseases, including epithelial skin cancer and acne. However, ATRA's cutaneous side effects, like redness and peeling, and its high instability limit its efficacy. To address these drawbacks and to improve ATRA solubilization, we prepared ATRA-loaded micelles (ATRA-TPGSs), by its encapsulation in D-α-tocopheryl-polyethylene-glycol-succinate (TPGS). First, to explore the feasibility of the project, a solubility study based on the equilibrium method was performed; then, six ATRA-TPGS formulations were prepared by the solvent-casting method using different TPGS amounts. ATRA-TPGSs showed small sizes (11-20 nm), low polydispersity, slightly negative zeta potential, and proved good encapsulation efficiency, confirmed by a chemometric-assisted Fourier transform infrared spectroscopy (FTIR) investigation. ATRA-TPGS stability was also investigated to choose the most stable formulation. Using Carbopol® 980 as gelling agent, ATRA-TPGS-loaded gels were obtained and analyzed for their rheological profiles. Ex vivo release studies from ATRA-TPGSs were performed by Franz cells, demonstrating a permeation after 24 h of 22 ± 4 µ cm-2. ATRA-TPGSs showed enhanced cytotoxic effects on melanoma cells, suggesting that these formulations may represent a valid alternative to improve patient compliance and to achieve more efficacious therapeutic outcomes.
Keywords: TPGS; drug delivery systems; micelles; nanocarrier-loaded gels; nanocarriers; retinoic acid; topical application.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease.Pharmaceutics. 2021 Jul 23;13(8):1128. doi: 10.3390/pharmaceutics13081128. Pharmaceutics. 2021. PMID: 34452090 Free PMC article.
-
Superiority of TPGS-loaded micelles in the brain delivery of vinpocetine via administration of thermosensitive intranasal gel.Int J Nanomedicine. 2019 Jul 23;14:5555-5567. doi: 10.2147/IJN.S213086. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31413562 Free PMC article.
-
Solubilized delivery of paliperidone palmitate by D-alpha-tocopheryl polyethylene glycol 1000 succinate micelles for improved short-term psychotic management.Drug Deliv. 2016;23(1):230-7. doi: 10.3109/10717544.2014.909907. Epub 2014 May 22. Drug Deliv. 2016. PMID: 24853962
-
Copolymers of poly(lactic acid) and D-α-tocopheryl polyethylene glycol 1000 succinate-based nanomedicines: versatile multifunctional platforms for cancer diagnosis and therapy.Expert Opin Drug Deliv. 2013 Apr;10(4):529-43. doi: 10.1517/17425247.2013.758632. Epub 2013 Jan 14. Expert Opin Drug Deliv. 2013. PMID: 23316695 Review.
-
Recent Advances in Vitamin E TPGS-Based Organic Nanocarriers for Enhancing the Oral Bioavailability of Active Compounds: A Systematic Review.Pharmaceutics. 2025 Apr 7;17(4):485. doi: 10.3390/pharmaceutics17040485. Pharmaceutics. 2025. PMID: 40284480 Free PMC article. Review.
Cited by
-
Successful Dendrimer and Liposome-Based Strategies to Solubilize an Antiproliferative Pyrazole Otherwise Not Clinically Applicable.Nanomaterials (Basel). 2022 Jan 11;12(2):233. doi: 10.3390/nano12020233. Nanomaterials (Basel). 2022. PMID: 35055251 Free PMC article.
-
Increased Water-Solubility and Maintained Antioxidant Power of Resveratrol by Its Encapsulation in Vitamin E TPGS Micelles: A Potential Nutritional Supplement for Chronic Liver Disease.Pharmaceutics. 2021 Jul 23;13(8):1128. doi: 10.3390/pharmaceutics13081128. Pharmaceutics. 2021. PMID: 34452090 Free PMC article.
-
Last Fifteen Years of Nanotechnology Application with Our Contribute.Nanomaterials (Basel). 2025 Feb 10;15(4):265. doi: 10.3390/nano15040265. Nanomaterials (Basel). 2025. PMID: 39997828 Free PMC article. Review.
-
Mutual Jellification of Two Bactericidal Cationic Polymers: Synthesis and Physicochemical Characterization of a New Two-Component Hydrogel.Pharmaceutics. 2022 Nov 11;14(11):2444. doi: 10.3390/pharmaceutics14112444. Pharmaceutics. 2022. PMID: 36432635 Free PMC article.
-
A Self-Forming Hydrogel from a Bactericidal Copolymer: Synthesis, Characterization, Biological Evaluations and Perspective Applications.Int J Mol Sci. 2022 Dec 1;23(23):15092. doi: 10.3390/ijms232315092. Int J Mol Sci. 2022. PMID: 36499417 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

