The Effects of Time-Restricted Eating versus Standard Dietary Advice on Weight, Metabolic Health and the Consumption of Processed Food: A Pragmatic Randomised Controlled Trial in Community-Based Adults
- PMID: 33807102
- PMCID: PMC8004978
- DOI: 10.3390/nu13031042
The Effects of Time-Restricted Eating versus Standard Dietary Advice on Weight, Metabolic Health and the Consumption of Processed Food: A Pragmatic Randomised Controlled Trial in Community-Based Adults
Abstract
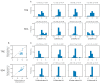
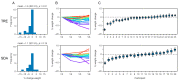
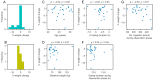
Weight loss is key to controlling the increasing prevalence of metabolic syndrome (MS) and its components, i.e., central obesity, hypertension, prediabetes and dyslipidaemia. The goals of our study were two-fold. First, we characterised the relationships between eating duration, unprocessed and processed food consumption and metabolic health. During 4 weeks of observation, 213 adults used a smartphone application to record food and drink consumption, which was annotated for food processing levels following the NOVA classification. Low consumption of unprocessed food and low physical activity showed significant associations with multiple MS components. Second, in a pragmatic randomised controlled trial, we compared the metabolic benefits of 12 h time-restricted eating (TRE) to standard dietary advice (SDA) in 54 adults with an eating duration > 14 h and at least one MS component. After 6 months, those randomised to TRE lost 1.6% of initial body weight (SD 2.9, p = 0.01), compared to the absence of weight loss with SDA (-1.1%, SD 3.5, p = 0.19). There was no significant difference in weight loss between TRE and SDA (between-group difference -0.88%, 95% confidence interval -3.1 to 1.3, p = 0.43). Our results show the potential of smartphone records to predict metabolic health and highlight that further research is needed to improve individual responses to TRE such as a shorter eating window or its actual clock time.
Keywords: NOVA classification; dietary advice; eating pattern; metabolic syndrome; processed food; time-restricted eating; weight loss.
Conflict of interest statement
The authors have no potential conflict of interest to declare. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
-
- Alberti K.G.M.M., Eckel R.H., Grundy S.M., Zimmet P.Z., Cleeman J.I., Donato K.A., Fruchart J.-C., James W.P.T., Loria C.M., Smith S.C., et al. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–1645. doi: 10.1161/circulationaha.109.192644. - DOI - PubMed
-
- Khera R., Murad M.H., Chandar A.K., Dulai P.S., Wang Z., Prokop L.J., Loomba R., Camilleri M., Singh S. Association of Pharmacological Treatments for Obesity With Weight Loss and Adverse Events: A Systematic Review and Meta-Analysis. JAMA. 2016;315:2424–2434. doi: 10.1001/jama.2016.7602. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

