Hepatic Stellate Cell Activation and Inactivation in NASH-Fibrosis-Roles as Putative Treatment Targets?
- PMID: 33807461
- PMCID: PMC8066583
- DOI: 10.3390/biomedicines9040365
Hepatic Stellate Cell Activation and Inactivation in NASH-Fibrosis-Roles as Putative Treatment Targets?
Abstract
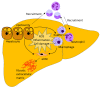
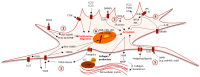
Hepatic fibrosis is the primary predictor of mortality in patients with non-alcoholic steatohepatitis (NASH). In this process, the activated hepatic stellate cells (HSCs) constitute the principal cells responsible for the deposition of a fibrous extracellular matrix, thereby driving the hepatic scarring. HSC activation, migration, and proliferation are controlled by a complex signaling network involving growth factors, lipotoxicity, inflammation, and cellular stress. Conversely, the clearance of activated HSCs is a prerequisite for the resolution of the extracellular fibrosis. Hence, pathways regulating the fate of the HSCs may represent attractive therapeutic targets for the treatment and prevention of NASH-associated hepatic fibrosis. However, the development of anti-fibrotic drugs for NASH patients has not yet resulted in clinically approved therapeutics, underscoring the complex biology and challenges involved when targeting the intricate cellular signaling mechanisms. This narrative review investigated the mechanisms of activation and inactivation of HSCs with a focus on NASH-associated hepatic fibrosis. Presenting an updated overview, this review highlights key cellular pathways with potential value for the development of future treatment modalities.
Keywords: HSC activation; HSC inactivation; fibrosis; hepatic stellate cells; non-alcoholic fatty liver disease; non-alcoholic steatohepatitis.
Conflict of interest statement
D.H.I. is an employee at Novo Nordisk A/S, a company involved in developing new therapies within NASH. A.Z. and P.T.-N. Declare no conflict of interest.
Figures

References
-
- Taylor R.S., Taylor R.J., Bayliss S., Hagström H., Nasr P., Schattenberg J.M., Ishigami M., Toyoda H., Wai-Sun Wong V., Peleg N., et al. Association Between Fibrosis Stage and Outcomes of Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Gastroenterology. 2020;158:1611–1625.e1612. doi: 10.1053/j.gastro.2020.01.043. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

