Impact of BMI on Survival Outcomes of Immunotherapy in Solid Tumors: A Systematic Review
- PMID: 33807855
- PMCID: PMC7961496
- DOI: 10.3390/ijms22052628
Impact of BMI on Survival Outcomes of Immunotherapy in Solid Tumors: A Systematic Review
Abstract
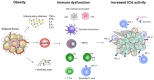
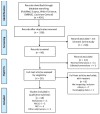
Growing research has focused on obesity as a prognostic factor during therapy with immune-checkpoint inhibitors (ICIs). The role of body-mass index (BMI) in predicting response and toxicity to ICIs is not clear, as studies have shown inconsistent results and significant interpretation biases. We performed a systematic review to evaluate the relationship between BMI and survival outcomes during ICIs, with a side focus on the incidence of immune-related adverse events (irAEs). A total of 17 studies were included in this systematic review. Altogether, the current evidence does not support a clearly positive association of BMI with survival outcomes. Regarding toxicities, available studies confirm a superimposable rate of irAEs among obese and normal weight patients. Intrinsic limitations of the analyzed studies include the retrospective nature, the heterogeneity of patients' cohorts, and differences in BMI categorization for obese patients across different studies. These factors might explain the heterogeneity of available results, and the subsequent absence of a well-established role of baseline BMI on the efficacy of ICIs among cancer patients. Further prospective studies are needed, in order to clarify the role of obesity in cancer patients treated with immunotherapy.
Keywords: anti-CTLA4; anti-PD1; anti-PDL1; cancer; immunotherapy; irAEs; obesity; obesity paradox; survival.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Association between body mass index and survival outcomes for cancer patients treated with immune checkpoint inhibitors: a systematic review and meta-analysis.J Transl Med. 2020 Jun 12;18(1):235. doi: 10.1186/s12967-020-02404-x. J Transl Med. 2020. PMID: 32532255 Free PMC article.
-
Association between immune-related side effects and efficacy and benefit of immune checkpoint inhibitors - A systematic review and meta-analysis.Cancer Treat Rev. 2021 Jan;92:102134. doi: 10.1016/j.ctrv.2020.102134. Epub 2020 Dec 3. Cancer Treat Rev. 2021. PMID: 33302134
-
Another side of the association between body mass index (BMI) and clinical outcomes of cancer patients receiving programmed cell death protein-1 (PD-1)/ Programmed cell death-ligand 1 (PD-L1) checkpoint inhibitors: A multicentre analysis of immune-related adverse events.Eur J Cancer. 2020 Mar;128:17-26. doi: 10.1016/j.ejca.2019.12.031. Epub 2020 Mar 5. Eur J Cancer. 2020. PMID: 32109847
-
Effect of body mass index on immune checkpoint inhibitor efficacy in patients with advanced cancer.Tumori. 2024 Dec;110(6):437-442. doi: 10.1177/03008916241291989. Epub 2024 Nov 6. Tumori. 2024. PMID: 39506383
-
Association between immune-related adverse events and long-term survival outcomes in patients treated with immune checkpoint inhibitors.Eur J Cancer. 2020 Jun;132:61-70. doi: 10.1016/j.ejca.2020.03.017. Epub 2020 Apr 22. Eur J Cancer. 2020. PMID: 32334337
Cited by
-
Nutritional risk index predicts the prognosis of gastric cancer patients with pyloric stenosis who received preoperative parenteral nutrition.Oncol Lett. 2023 Jul 31;26(3):401. doi: 10.3892/ol.2023.13988. eCollection 2023 Sep. Oncol Lett. 2023. PMID: 37600343 Free PMC article.
-
Nutritional Risk Index (NRI) predicts the clinical outcomes of patients with gastric cancer who received immune checkpoint inhibitors (PD-1/PD-L1).Medicine (Baltimore). 2025 Jan 3;104(1):e40898. doi: 10.1097/MD.0000000000040898. Medicine (Baltimore). 2025. PMID: 40184134 Free PMC article.
-
Potential roles of sex-linked differences in obesity and cancer immunotherapy: revisiting the obesity paradox.NPJ Metab Health Dis. 2024;2(1):5. doi: 10.1038/s44324-024-00007-4. Epub 2024 May 23. NPJ Metab Health Dis. 2024. PMID: 38800540 Free PMC article. Review.
-
Clinical variables associated with immune checkpoint inhibitor outcomes in patients with metastatic urothelial carcinoma: a multicentre retrospective cohort study.BMJ Open. 2024 Mar 29;14(3):e081480. doi: 10.1136/bmjopen-2023-081480. BMJ Open. 2024. PMID: 38553056 Free PMC article.
-
Nonlinear association between body mass index and overall survival in advanced NSCLC patients treated with immune checkpoint blockade.Cancer Immunol Immunother. 2023 May;72(5):1225-1232. doi: 10.1007/s00262-022-03320-3. Epub 2022 Nov 16. Cancer Immunol Immunother. 2023. PMID: 36383245 Free PMC article.
References
-
- Migden M.R., Khushalani N.I., Chang A.L.S., Lewis K.D., Schmults C.D., Hernandez-Aya L., Meier F., Schadendorf D., Guminski A., Haushild A., et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: Results from an open-label, phase 2, single-arm trial. Lancet Oncol. 2020;21:294–305. doi: 10.1016/S1470-2045(19)30728-4. - DOI - PMC - PubMed
-
- Horn L., Spigel D.R., Vokes E.E., Holgado E., Ready N., Steins M., Poddubskaya E., Borghaei H., Felip E., Paz-Ares L., et al. Nivolumab Versus Docetaxel in Previously Treated Patients With Advanced Non-Small-Cell Lung Cancer: Two-Year Outcomes From Two Randomized, Open-Label, Phase III Trials (CheckMate 017 and CheckMate 057) J. Clin. Oncol. 2017;35:3924–3933. doi: 10.1200/JCO.2017.74.3062. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

