Ionic Liquid-Based Surfactants: Recent Advances in Their Syntheses, Solution Properties, and Applications
- PMID: 33808369
- PMCID: PMC8036849
- DOI: 10.3390/polym13071100
Ionic Liquid-Based Surfactants: Recent Advances in Their Syntheses, Solution Properties, and Applications
Abstract
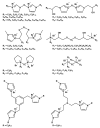
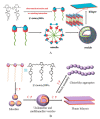
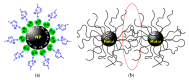
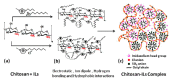
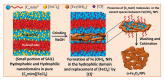
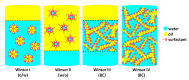
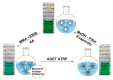
The impetus for the expanding interest in ionic liquids (ILs) is their favorable properties and important applications. Ionic liquid-based surfactants (ILBSs) carry long-chain hydrophobic tails. Two or more molecules of ILBSs can be joined by covalent bonds leading, e.g., to gemini compounds (GILBSs). This review article focuses on aspects of the chemistry and applications of ILBSs and GILBSs, especially in the last ten years. Data on their adsorption at the interface and micelle formation are relevant for the applications of these surfactants. Therefore, we collected data for 152 ILBSs and 11 biamphiphilic compounds. The head ions of ILBSs are usually heterocyclic (imidazolium, pyridinium, pyrrolidinium, etc.). Most of these head-ions are also present in the reported 53 GILBSs. Where possible, we correlate the adsorption/micellar properties of the surfactants with their molecular structures, in particular, the number of carbon atoms present in the hydrocarbon "tail". The use of ILBSs as templates for the fabrication of mesoporous nanoparticles enables better control of particle porosity and size, hence increasing their usefulness. ILs and ILBSs form thermodynamically stable water/oil and oil/water microemulsions. These were employed as templates for (radical) polymerization reactions, where the monomer is the "oil" component. The formed polymer nanoparticles can be further stabilized against aggregation by using a functionalized ILBS that is co-polymerized with the monomers. In addition to updating the literature on the subject, we hope that this review highlights the versatility and hence the potential applications of these classes of surfactants in several fields, including synthesis, catalysis, polymers, decontamination, and drug delivery.
Keywords: adsorption at water/air interface; catalysis; drug delivery; formation of micelles and microemulsions; gemini ionic liquid-based surfactants; ionic liquid-based surfactants; ionic liquids; mesoporous nanoparticles; molecular structure/properties relationships; polymerization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures















Similar articles
-
Gemini ionic liquid-based surfactants: efficient synthesis, surface activity, and use as inducers for the fabrication of Cu2O nanoparticles.RSC Adv. 2023 Oct 24;13(44):31128-31140. doi: 10.1039/d3ra04646j. eCollection 2023 Oct 18. RSC Adv. 2023. PMID: 37881753 Free PMC article.
-
Aggregation behavior and antimicrobial activity of ester-functionalized imidazolium- and pyridinium-based ionic liquids in aqueous solution.Langmuir. 2013 Feb 26;29(8):2536-45. doi: 10.1021/la304752e. Epub 2013 Feb 11. Langmuir. 2013. PMID: 23360222
-
Surface active ionic liquids: study of the micellar properties of 1-(1-alkyl)-3-methylimidazolium chlorides and comparison with structurally related surfactants.J Colloid Interface Sci. 2011 Sep 1;361(1):186-94. doi: 10.1016/j.jcis.2011.04.108. Epub 2011 May 10. J Colloid Interface Sci. 2011. PMID: 21641609
-
Scrutinizing Self-Assembly, Surface Activity and Aggregation Behavior of Mixtures of Imidazolium Based Ionic Liquids and Surfactants: A Comprehensive Review.Front Chem. 2021 May 13;9:667941. doi: 10.3389/fchem.2021.667941. eCollection 2021. Front Chem. 2021. PMID: 34055738 Free PMC article. Review.
-
Gemini and Bicephalous Surfactants: A Review on Their Synthesis, Micelle Formation, and Uses.Int J Mol Sci. 2022 Feb 4;23(3):1798. doi: 10.3390/ijms23031798. Int J Mol Sci. 2022. PMID: 35163721 Free PMC article. Review.
Cited by
-
Synthesis and evaluation of amino acid ionic liquid for enhanced oil recovery: experimental and modeling simulation studies.Sci Rep. 2025 Jan 16;15(1):2201. doi: 10.1038/s41598-025-85560-8. Sci Rep. 2025. PMID: 39820535 Free PMC article.
-
Gemini ionic liquid-based surfactants: efficient synthesis, surface activity, and use as inducers for the fabrication of Cu2O nanoparticles.RSC Adv. 2023 Oct 24;13(44):31128-31140. doi: 10.1039/d3ra04646j. eCollection 2023 Oct 18. RSC Adv. 2023. PMID: 37881753 Free PMC article.
-
Nanomicellar Extraction of Polyphenols-Methodology and Applications Review.Int J Mol Sci. 2021 Oct 21;22(21):11392. doi: 10.3390/ijms222111392. Int J Mol Sci. 2021. PMID: 34768823 Free PMC article. Review.
-
Review on Amphiphilic Ionic Liquids as New Surfactants: From Fundamentals to Applications.Top Curr Chem (Cham). 2021 Nov 29;380(1):5. doi: 10.1007/s41061-021-00362-6. Top Curr Chem (Cham). 2021. PMID: 34842981 Review.
-
Ionic Liquids in Pharmaceutical and Biomedical Applications: A Review.Pharmaceutics. 2024 Jan 22;16(1):151. doi: 10.3390/pharmaceutics16010151. Pharmaceutics. 2024. PMID: 38276519 Free PMC article. Review.
References
-
- Pino V., Germán-Hernández M., Martín-Pérez A., Anderson J.L. Ionic Liquid-Based Surfactants in Separation Science. Sep. Sci. Technol. 2012;47:264–276. doi: 10.1080/01496395.2011.620589. - DOI
-
- Cao H., Hu Y., Xu W., Wang Y., Guo X. Recent Progress in the Assembly Behavior of Imidazolium-Based Ionic Liquid Surfactants. J. Mol. Liq. 2020;319:114354. doi: 10.1016/j.molliq.2020.114354. - DOI
-
- Hejazifar M., Lanaridi O., Bica-Schröder K. Ionic Liquid Based Microemulsions: A Review. J. Mol. Liq. 2020;303:112264. doi: 10.1016/j.molliq.2019.112264. - DOI
-
- Lee D.J. Enthalpy-Entropy Compensation in Ionic Micelle Formation. Colloid Polym. Sci. 1995;273:539–543. doi: 10.1007/BF00658683. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

