Development and Translation of NanoBEO, a Nanotechnology-Based Delivery System of Bergamot Essential Oil Deprived of Furocumarins, in the Control of Agitation in Severe Dementia
- PMID: 33809385
- PMCID: PMC7999378
- DOI: 10.3390/pharmaceutics13030379
Development and Translation of NanoBEO, a Nanotechnology-Based Delivery System of Bergamot Essential Oil Deprived of Furocumarins, in the Control of Agitation in Severe Dementia
Abstract
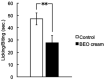
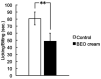
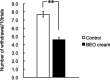
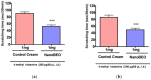
Dementia is one of the most common causes of disability worldwide characterized by memory loss, cognitive impairment, and behavioral and psychological symptoms (BPSD), including agitation. Treatment of the latter consists of the off-label use of harmful atypical antipsychotics, though a significant reduction is afforded by pain control. The use of an essential oil endowed with analgesic properties and devoid of toxicity would represent an important option for the management of agitation in dementia. Therefore, the aim of this study was to engineer a nanotechnology delivery system based on solid lipid nanoparticles loaded with bergamot essential oil (BEO) and devised in the pharmaceutical form of an odorless cream (NanoBEO) to confirm its analgesic efficacy for further development and application to control agitation in dementia. BEO has proven strong antinociceptive and anti-allodynic properties and, in its bergapten-free form, it is completely devoid of phototoxicity. NanoBEO has been studied in vivo confirming the previously reported analgesic activity of BEO to which is now added its anti-itching properties. Due to the nanotechnology delivery system, the stability of titrated BEO components is guaranteed. Finally, the latter invention, currently under patent consideration, is smell-devoid allowing efficacy and safety to be established in double-blind clinical trials; until now the latter studies have been impeded in aromatherapy by the strong odor of essential oils. A clinical trial NCT04321889 has been designed to provide information about the efficacy and safety of NanoBEO on agitation and pain in patients suffering from severe dementia.
Keywords: BPSD; agitation; dementia; essential oil of bergamot; nanotechnology delivery system; pain.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Winblad B., Amouyel P., Andrieu S., Ballard C., Brayne C., Brodaty H., Cedazo-Minguez A., Dubois B., Edvardsson D., Feldman H., et al. Defeating Alzheimer’s disease and other dementias: A priority for European science and society. Lancet Neurol. 2016;15:455–532. doi: 10.1016/S1474-4422(16)00062-4. - DOI - PubMed
-
- Scuteri D., Rombola L., Tridico L., Mizoguchi H., Watanabe C., Sakurada T., Sakurada S., Corasaniti M.T., Bagetta G., Morrone L.A. Neuropharmacological Properties of the Essential Oil of Bergamot for the Clinical Management of Pain-Related BPSDs. Curr. Med. Chem. 2019;26:3764–3774. doi: 10.2174/0929867325666180307115546. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

