Could Small Heat Shock Protein HSP27 Be a First-Line Target for Preventing Protein Aggregation in Parkinson's Disease?
- PMID: 33809767
- PMCID: PMC8002365
- DOI: 10.3390/ijms22063038
Could Small Heat Shock Protein HSP27 Be a First-Line Target for Preventing Protein Aggregation in Parkinson's Disease?
Abstract
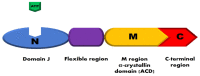
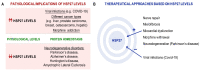
Small heat shock proteins (HSPs), such as HSP27, are ubiquitously expressed molecular chaperones and are essential for cellular homeostasis. The major functions of HSP27 include chaperoning misfolded or unfolded polypeptides and protecting cells from toxic stress. Dysregulation of stress proteins is associated with many human diseases including neurodegenerative diseases, such as Parkinson's disease (PD). PD is characterized by the presence of aggregates of α-synuclein in the central and peripheral nervous system, which induces the degeneration of dopaminergic neurons in the substantia nigra pars compacta (SNpc) and in the autonomic nervous system. Autonomic dysfunction is an important non-motor phenotype of PD, which includes cardiovascular dysregulation, among others. Nowadays, the therapies for PD focus on dopamine (DA) replacement. However, certain non-motor symptoms with a great impact on quality of life do not respond to dopaminergic drugs; therefore, the development and testing of new treatments for non-motor symptoms of PD remain a priority. Since small HSP27 was shown to prevent α-synuclein aggregation and cytotoxicity, this protein might constitute a suitable target to prevent or delay the motor and non-motor symptoms of PD. In the first part of our review, we focus on the cardiovascular dysregulation observed in PD patients. In the second part, we present data on the possible role of HSP27 in preventing the accumulation of amyloid fibrils and aggregated forms of α-synuclein. We also include our own studies, highlighting the possible protective cardiac effects induced by L-DOPA treatment through the enhancement of HSP27 levels and activity.
Keywords: L-DOPA; Parkinson´s disease; cardiac dysfunction; non-motor symptoms; small heat shock protein 27; α-synuclein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Linquistm S. The heat-shock response. Ann. Int. Rev. Biochem. 1986;55:1151–1191. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

