Immune response characterization in a human challenge study with a Shigella flexneri 2a bioconjugate vaccine
- PMID: 33813141
- PMCID: PMC8047506
- DOI: 10.1016/j.ebiom.2021.103308
Immune response characterization in a human challenge study with a Shigella flexneri 2a bioconjugate vaccine
Abstract
Background: Diarrheal diseases are a leading cause of global morbidity and mortality affecting all ages, but especially children under the age of five in resource-limited settings. Shigella is a leading contributor to diarrheal diseases caused by bacterial pathogens and is considered a significant antimicrobial resistance threat. While improvements in hygiene, and access to clean water help as control measures, vaccination remains one of the most viable options for significantly reducing morbidity and mortality.
Methods: Flexyn2a is a bioconjugate vaccine manufactured using novel conjugation methodologies enzymatically linking the O-polysaccharide of S. flexneri 2a to exotoxin A of Pseudomonas aeruginosa. The protective capacity of Flexyn2a was assessed in a controlled human infection model after two intramuscular immunizations. Immune responses pre- and post-immunization and/or infection were investigated and are described here.
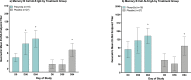
Findings: Flexyn2a induced lipopolysaccharide (LPS)-specific serum IgG responses post-immunization which were associated with protection against shigellosis. Additionally, several other immune parameters, including memory B cell responses, bactericidal antibodies and serum IgA, were also elevated in vaccinees protected against shigellosis. Immunization with Flexyn2a also induced gut-homing, LPS-specific IgG and IgA secreting B cells, indicating the vaccine induced immune effectors functioning at the site of intestinal infection.
Interpretation: Collectively, the results of these immunological investigations provide insights into protective immune mechanisms post-immunization with Flexyn2a which can be used to further guide vaccine development and may have applicability to the larger Shigella vaccine field.
Funding: Funding for this study was provided through a Wellcome Trust grant.
Keywords: Bioconjugate vaccine; Gut-homing responses; Human challenge; Immunogenicity; Parenteral immunization; Shigella.
Published by Elsevier B.V.
Conflict of interest statement
Declaration of Competing Interest CA, ALB, JB, KAC, BD, AMD, DE, BF, RF, VGF, JH, RWK, PM, DS, KRT, HPW report grants from Wellcome Trust during the conduct of the study. SC, CKP, MSR report no conflicts of interest for any of the materials presented in the manuscript.
Figures




Comment in
-
Shigella conjugate vaccine efficacy trial in controlled human model and potential immune correlates of protection.EBioMedicine. 2021 Apr;66:103343. doi: 10.1016/j.ebiom.2021.103343. Epub 2021 Apr 16. EBioMedicine. 2021. PMID: 33873142 Free PMC article. No abstract available.
References
-
- BCG. Vaccines to tackle drug resistant infections: An evaluation of R&D opportunities: Wellcome Trust & Boston Consulting Group, https://vaccinesforamr.org/wp-content/uploads/2018/09/Vaccines_for_AMR.pdf. 2018.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

