Inhibition of PKCε induces primordial germ cell reprogramming into pluripotency by HIF1&2 upregulation and histone acetylation
- PMID: 33815934
- PMCID: PMC8012778
Inhibition of PKCε induces primordial germ cell reprogramming into pluripotency by HIF1&2 upregulation and histone acetylation
Abstract
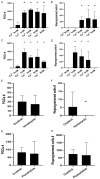
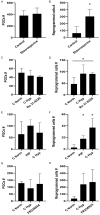
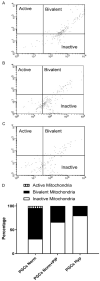
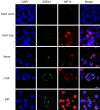
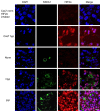
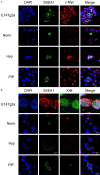
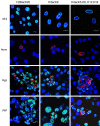
Historically, primordial germ cells (PGCs) have been a good model to study pluripotency. Despite their low numbers and limited accessibility in the mouse embryo, they can be easily and rapidly reprogrammed at high efficiency with external physicochemical factors and do not require transcription factor transfection. Employing this model to deepen our understanding of cell reprogramming, we specifically aimed to determine the relevance of Ca2+ signal transduction pathway components in the reprogramming process. Our results showed that PGC reprogramming requires a normal extracellular [Ca2+] range, in contrast to neoplastic or transformed cells, which can continue to proliferate in Ca2+-deficient media, differentiating normal reprogramming from neoplastic transformation. Our results also showed that a spike in extracellular [Ca2+] of 1-3 mM can directly reprogram PGC. Intracellular manipulation of Ca2+ signal transduction pathway components revealed that inhibition of classical Ca2+ and diacylglycerol (DAG)-dependent PKCs, or intriguingly, of only the novel DAG-dependent PKC, PKCε, were able to induce reprogramming. PKCε inhibition changed the metabolism of PGCs toward glycolysis, increasing the proportion of inactive mitochondria. This metabolic switch from oxidative phosphorylation to glycolysis is mediated by hypoxia-inducible factors (HIFs), given we found upregulation of both HIF1α and HIF2α in the first 48 hours of culturing. PKCε inhibition did not change the classical pluripotency gene expression of PGCs, Oct4, or Nanog. PKCε inhibition changed the histone acetylation of PGCs, with histones H2B, H3, and H4 becoming acetylated in PKCε-inhibited cultures (markers were H2BacK20, H3acK9, and H4acK5K8, K12, K16), suggesting that reprogramming by PKCε inhibition is mediated by histone acetylation.
Keywords: Cellular reprograming; HIF; PKC; calcium signaling; histone acetylation; pluripotency; primordial germ cells.
AJSC Copyright © 2021.
Conflict of interest statement
None.
Figures

Similar articles
-
Metabolic Reprogramming, Autophagy, and Reactive Oxygen Species Are Necessary for Primordial Germ Cell Reprogramming into Pluripotency.Oxid Med Cell Longev. 2017;2017:4745252. doi: 10.1155/2017/4745252. Epub 2017 Jul 5. Oxid Med Cell Longev. 2017. PMID: 28757909 Free PMC article.
-
Hypoxia induces pluripotency in primordial germ cells by HIF1α stabilization and Oct4 deregulation.Antioxid Redox Signal. 2015 Jan 20;22(3):205-23. doi: 10.1089/ars.2014.5871. Epub 2014 Oct 30. Antioxid Redox Signal. 2015. PMID: 25226357
-
Shortened G1 phase of cell cycle and decreased histone H3K27 methylation are associated with AKT-induced enhancement of primordial germ cell reprogramming.Dev Growth Differ. 2019 Aug;61(6):357-364. doi: 10.1111/dgd.12621. Epub 2019 Jun 14. Dev Growth Differ. 2019. PMID: 31199000
-
In or out stemness: comparing growth factor signalling in mouse embryonic stem cells and primordial germ cells.Curr Stem Cell Res Ther. 2009 May;4(2):87-97. doi: 10.2174/157488809788167391. Curr Stem Cell Res Ther. 2009. PMID: 19442193 Review.
-
Reprogramming of germ cells into pluripotency.World J Stem Cells. 2016 Aug 26;8(8):251-9. doi: 10.4252/wjsc.v8.i8.251. World J Stem Cells. 2016. PMID: 27621759 Free PMC article. Review.
Cited by
-
PKC inhibitors regulate stem cell self-renewal by regulating H3K27me3 and H3K9me3.Am J Transl Res. 2022 Jun 15;14(6):4295-4309. eCollection 2022. Am J Transl Res. 2022. PMID: 35836851 Free PMC article.
-
Hypoxia Increases the Efficiencies of Cellular Reprogramming and Oncogenic Transformation in Human Blood Cell Subpopulations In Vitro and In Vivo.Cells. 2024 Jun 4;13(11):971. doi: 10.3390/cells13110971. Cells. 2024. PMID: 38891103 Free PMC article.
-
Early Gonadal Development and Sex Determination in Mammal.Int J Mol Sci. 2022 Jul 6;23(14):7500. doi: 10.3390/ijms23147500. Int J Mol Sci. 2022. PMID: 35886859 Free PMC article. Review.
-
Changes in lipid profile and SOX-2 expression in RM-1 cells after co-culture with preimplantation embryos or with deproteinated blastocyst extracts.Mol Omics. 2022 Jul 11;18(6):480-489. doi: 10.1039/d2mo00071g. Mol Omics. 2022. PMID: 35506630 Free PMC article.
-
Low-input and single-cell methods for Infinium DNA methylation BeadChips.Nucleic Acids Res. 2024 Apr 24;52(7):e38. doi: 10.1093/nar/gkae127. Nucleic Acids Res. 2024. PMID: 38407446 Free PMC article.
References
-
- De Miguel MP, Alcaina Y, de la Maza DS. Primordial germ cell reprogramming. In: Germ Cell. IntechOpen; 2018. pp. 43–62.
-
- López-Iglesias P, Alcaina Y, Tapia N, Sabour D, Arauzo-Bravo MJ, Sainz De La Maza D, Berra E, O’Mara AN, Nistal M, Ortega S, Donovan PJ, Schöler HR, De Miguel MP. Hypoxia induces pluripotency in primordial germ cells by HIF1α stabilization and Oct4 deregulation. Antioxid Redox Signal. 2015;22:205–223. - PubMed
-
- Resnick JL, Bixler LS, Cheng L, Donovan PJ. Long-term proliferation of mouse primordial germ cells in culture. Nature. 1992;359:550–551. - PubMed
-
- Matsui Y, Zsebo K, Hogan BL. Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell. 1992;70:841–847. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
