Illustration of a number of atypical computed tomography manifestations of active pulmonary tuberculosis
- PMID: 33816198
- PMCID: PMC7930668
- DOI: 10.21037/qims-20-1323
Illustration of a number of atypical computed tomography manifestations of active pulmonary tuberculosis
Abstract
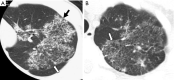
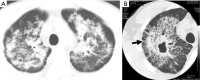
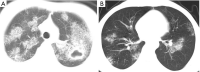
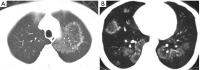
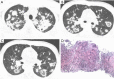
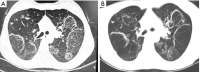
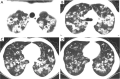
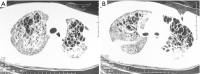
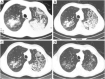
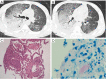
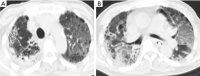
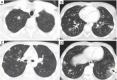
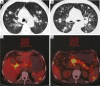
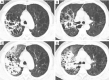
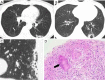
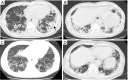
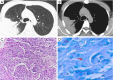
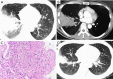
Tuberculosis is a serious public health challenge facing mankind and one of the top ten causes of death. Diagnostic imaging plays an important role, particularly for the diagnosis and treatment planning of tuberculosis patients with negative microbiology results. This article illustrates a number of atypical computed tomography (CT) appearances of pulmonary tuberculosis (PTB), including (I) clustered micronodules (CMNs) sign; (II) reversed halo sign (RHS); (III) tuberculous pneumatocele; (IV) hematogenously disseminated PTB with predominantly diffuse ground glass opacity manifestation; (V) hematogenously disseminated PTB with randomly distributed non-miliary nodules; (VI) PTB changes occur on the background of emphysema or honeycomb changes of interstitial pneumonia; and (VII) PTB manifesting as organizing pneumonia. While the overall incidence of PTB is decreasing globally, the incidence of atypical manifestations of tuberculosis is increasing. A good understanding of the atypical CT imaging changes of active PTB shall help the diagnosis and differential diagnosis of PTB in clinical practice.
Keywords: Pulmonary tuberculosis (PTB); cluster of micronodules; emphysema; interstitial pneumonia; miliary tuberculosis; organizing pneumonia (OP); pneumatocele; reversed halo sign (RHS); sarcoid galaxy sign.
2021 Quantitative Imaging in Medicine and Surgery. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/qims-20-1323). YXJW serves as an Editor-in-Chief of Quantitative Imaging in Medicine and Surgery. The authors have no other conflicts of interest to declare.
Figures


References
-
- World Health Organization. Global tuberculosis report 2018. Available online: www.who.int/tb/publications/global_report (accessed 15 March 2019).
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
