No functional TRPA1 in cardiomyocytes
- PMID: 33819369
- PMCID: PMC11478933
- DOI: 10.1111/apha.13659
No functional TRPA1 in cardiomyocytes
Abstract
Aim: There is mounting evidence that TRPA1 has a role in cardiac physiology and pathophysiology. We aim to clarify the site of TRPA1 expression in the heart and in particular whether the channel is expressed in cardiomyocytes.
Methods: Due to the high calcium conductance of TRPA1, and marginal calcium changes being detectable, microfluorimetry in primary mouse cardiomyocytes, and in the cardiomyocyte cell lines H9c2 and HL-1, was applied. TRPA1 mRNA in mouse and human hearts, primary cardiomyocytes, and the cardiac cell lines were quantified. Dorsal root ganglia served as control for both methods.
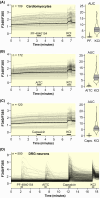
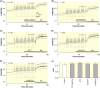
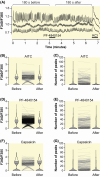
Results: In addition to AITC, the more potent and specific TRPA1 agonists JT010 and PF-4840154 failed to elicit a TRPA1-mediated response in native and electrically paced primary cardiomyocytes, and the cardiomyocyte cell lines H9c2 and HL-1. There were only marginal levels of TRPA1 mRNA in cardiomyocytes and cardiac cell lines, also in conditions of cell differentiation or inflammation, which might occur in pathophysiological conditions. Similarly, TRPV1 agonist capsaicin did not activate primary mouse cardiomyocytes, did not alter electrically paced activity in these, and did not activate H9c2 cells or alter spontaneous activity of HL-1 cells. Human pluripotent stem cells differentiated to cardiomyocytes had no relevant TRPA1 mRNA levels. Also in human post-mortem heart samples, TRPA1 mRNA levels were substantially lower compared with the respective dorsal root ganglion.
Conclusion: The results do not question a role of TRPA1 in the heart but exclude a direct effect in cardiomyocytes.
Keywords: TRPA1; TRPV1; cardiomyocytes; heart; human; mouse.
© 2021 The Authors. Acta Physiologica published by John Wiley & Sons Ltd on behalf of Scandinavian Physiological Society.
Conflict of interest statement
There is no conflict of interest.
Figures


Comment in
-
TRPA1 and TRPV1, do we hold you in our heart?Acta Physiol (Oxf). 2021 Aug;232(4):e13695. doi: 10.1111/apha.13695. Epub 2021 Jun 3. Acta Physiol (Oxf). 2021. PMID: 34041855 No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

