A quality improvement project to address the challenges surrounding zoledronic acid use in children
- PMID: 33825940
- PMCID: PMC8025437
- DOI: 10.1007/s00774-021-01214-5
A quality improvement project to address the challenges surrounding zoledronic acid use in children
Abstract
Introduction: Zoledronic acid (ZA) is an intravenous bisphosphonate used to treat pediatric osteoporosis. Adverse events including hypocalcemia and acute phase reaction (APR) are common following first-infusion. The purpose of this report is to describe implementation of a ZA clinical practice guideline and the subsequent process changes to improve adherence to aspects of the protocol related to safety and efficacy.
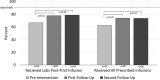
Methods: Quality assurance was evaluated by chart review over a 5-year period to compare the prevalence of hypocalcemia and APR to published data. A quality improvement (QI) initiative consisting of process changes including the addition of an endocrine RN to coordinate infusions and a shift to patient/family self-scheduling of infusions was conducted. The effect of the interventions on safety (completion of pre- and post-infusion bloodwork) and efficacy (receipt of all prescribed infusions) outcomes was evaluated.
Results: Seventy-two patients received 244 infusions over the period. The frequency of hypocalcemia (22%) and APR (31%) was consistent with prior reports. 99% of patients received pre-infusion bloodwork, 78% received post-first-infusion bloodwork, and 47% received all prescribed infusions. QI initiatives increased the percentage of patients receiving post-first-infusion bloodwork from 67 to 79% and those receiving all infusions from 62 to 74%, but fell short of the goal of 90%.
Conclusions: The implementation of a standardized protocol for ZA use in children was successful in confirming patient eligibility with pre-infusion bloodwork but failed to ensure that patients obtained post-first-infusion bloodwork and received all prescribed infusions. Further efforts to systematize the management of children on ZA are needed.
Keywords: Bisphosphonates; Osteoporosis; Quality assessment; Quality improvement; Zoledronic acid.
Conflict of interest statement
David R Weber, Maria Cristina Gil Diaz, Silvia Louis, Noya Rackovsky, Roman Rahmani, and Sierra D Stauber declare that they have no conflict of interest.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

