Neural stem cells traffic functional mitochondria via extracellular vesicles
- PMID: 33826607
- PMCID: PMC8055036
- DOI: 10.1371/journal.pbio.3001166
Neural stem cells traffic functional mitochondria via extracellular vesicles
Abstract
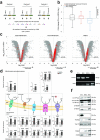
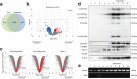
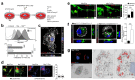
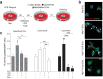
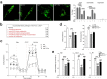
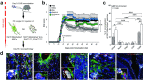
Neural stem cell (NSC) transplantation induces recovery in animal models of central nervous system (CNS) diseases. Although the replacement of lost endogenous cells was originally proposed as the primary healing mechanism of NSC grafts, it is now clear that transplanted NSCs operate via multiple mechanisms, including the horizontal exchange of therapeutic cargoes to host cells via extracellular vesicles (EVs). EVs are membrane particles trafficking nucleic acids, proteins, metabolites and metabolic enzymes, lipids, and entire organelles. However, the function and the contribution of these cargoes to the broad therapeutic effects of NSCs are yet to be fully understood. Mitochondrial dysfunction is an established feature of several inflammatory and degenerative CNS disorders, most of which are potentially treatable with exogenous stem cell therapeutics. Herein, we investigated the hypothesis that NSCs release and traffic functional mitochondria via EVs to restore mitochondrial function in target cells. Untargeted proteomics revealed a significant enrichment of mitochondrial proteins spontaneously released by NSCs in EVs. Morphological and functional analyses confirmed the presence of ultrastructurally intact mitochondria within EVs with conserved membrane potential and respiration. We found that the transfer of these mitochondria from EVs to mtDNA-deficient L929 Rho0 cells rescued mitochondrial function and increased Rho0 cell survival. Furthermore, the incorporation of mitochondria from EVs into inflammatory mononuclear phagocytes restored normal mitochondrial dynamics and cellular metabolism and reduced the expression of pro-inflammatory markers in target cells. When transplanted in an animal model of multiple sclerosis, exogenous NSCs actively transferred mitochondria to mononuclear phagocytes and induced a significant amelioration of clinical deficits. Our data provide the first evidence that NSCs deliver functional mitochondria to target cells via EVs, paving the way for the development of novel (a)cellular approaches aimed at restoring mitochondrial dysfunction not only in multiple sclerosis, but also in degenerative neurological diseases.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: SP is co-founder, CSO and shareholder (>5%) of CITC Ltd. and iSTEM Therapeutics Litd., and co-founder and Non-executive Director at asitia Therapeutics Ltd.; LPJ is shareholder of CITC Ltd.; JAS is a Project Manager and Senior Research Associate at CITC Ltd. and Director of Research of iSTEM Therapeutics Ltd.; BP is an employee of NanoFCM and his contributions to this paper were made as part of their employment.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
- MC_UU_12022/6/MRC_/Medical Research Council/United Kingdom
- MR/V011561/1/MRC_/Medical Research Council/United Kingdom
- MC_UP_1002/1/MRC_/Medical Research Council/United Kingdom
- 210688/Z/18/Z/WT_/Wellcome Trust/United Kingdom
- MC_EX_MR/P007031/1/MRC_/Medical Research Council/United Kingdom
- DH_/Department of Health/United Kingdom
- MR/K026682/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- MC_UP_1101/3/MRC_/Medical Research Council/United Kingdom
- 101835/Z/13/Z/WT_/Wellcome Trust/United Kingdom
- MRF-175-0001-RG-ZEVI-C0898/MRF_/MRF_/United Kingdom
- MRF-155-0002-RG-ZEVIA/MRF_/MRF_/United Kingdom
- RRAG/214/WT_/Wellcome Trust/United Kingdom
- MR/P008801/1/MRC_/Medical Research Council/United Kingdom
- MC_UU_00015/5/MRC_/Medical Research Council/United Kingdom
- MR/K026682/1/MRC_/Medical Research Council/United Kingdom
- RRZA/057 RG79423/WT_/Wellcome Trust/United Kingdom
- MC_UU_00015/8/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

