Meeting the Demand for Unrelated Donors in the Midst of the COVID-19 Pandemic: Rapid Adaptations by the National Marrow Donor Program and Its Network Partners Ensured a Safe Supply of Donor Products
- PMID: 33830022
- PMCID: PMC7834500
- DOI: 10.1016/j.jtct.2020.10.014
Meeting the Demand for Unrelated Donors in the Midst of the COVID-19 Pandemic: Rapid Adaptations by the National Marrow Donor Program and Its Network Partners Ensured a Safe Supply of Donor Products
Abstract
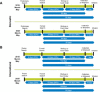
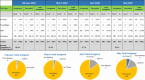
The impact of the coronavirus disease 2019 (COVID-19) pandemic on hematopoietic cell transplant (HCT) donor registries and transplant center (TC) practices is underreported. This article reports on the National Marrow Donor Program (NMDP) Be The Match Registry and its coordinating the provision of unrelated donor (URD) products to domestic and international TCs during the initial 3 months of the COVID-19 pandemic (March through May 2020). Specifically, NMDP data are presented for disease indications for transplant, URD search volumes and availability, graft requests and processing, courier utilization and performance, and conversion rates from formal donor search and workup to graft collection and shipment. Data following the onset of COVID-19 are compared to the immediate 3 months prior to the COVID-19 pandemic (December 2019 through February 2020) and the same quarter 1 year prior to COVID-19 (March through May 2019). During the initial onset of COVID-19 and compared to 1 year prior, TCs requested and the NMDP performed less donor searches. More multiple URD and direct to workup requests were processed by the NMDP, which likely reflected reductions in donor availability. Yet TCs continued to perform allogeneic transplants for acute disease indications like acute leukemia and myelodysplasia, using more cryopreserved grafts than before COVID-19. In comparison to prepandemic patient cycle conversion rates and durations, the NMDP was able to convert patient cycles at nearly the same or higher rates and in similar or shorter periods of time. Last, despite significant challenges caused by the pandemic, including interruptions in domestic courier services and travel restrictions, graft products were delivered to and received by TCs in similar periods of time than before COVID-19. Taken together, these data show that NMDP service line operations continued to function effectively during the early phases of the COVID-19 pandemic, ensuring requests for and delivery of URD products to domestic and international allogeneic HCT recipients.
Keywords: Bone marrow transplant; COVID-19; Coronavirus; Donor registry; Donor search; Graft; HLA matching; National Marrow Donor Program; Peripheral blood transplant; Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2); Umbilical cord blood; Unrelated donor.
Copyright © 2020 The American Society for Transplantation and Cellular Therapy. Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Miller I.F., Becker A.D., Grenfell B.T., Metcalf C.J.E. Disease and healthcare burden of COVID-19 in the United States. Nat Med. 2020;26:1212–1217. - PubMed
-
- Galea S., Abdalla S.M. COVID-19 pandemic, unemployment, and civil unrest: underlying deep racial and socioeconomic divides. JAMA. 2020;324:227–228. - PubMed
-
- Algwaiz G., Aljurf M., Koh M., et al. Real-world issues and potential solutions in hematopoietic cell transplantation during the COVID-19 pandemic: perspectives from the Worldwide Network for Blood and Marrow Transplantation and Center for International Blood and Marrow Transplant Research Health Services and International Studies Committee. Biol Blood Marrow Transplant. 2020;26:2181–2189. - PMC - PubMed
-
- Ljungman P., Mikulska M., de la Camara R., et al. Correction: The challenge of COVID-19 and hematopoietic cell transplantation: EBMT recommendations for management of hematopoietic cell transplant recipients, their donors, and patients undergoing CAR T-cell therapy [e-pub ahead of print]. Bone Marrow Transplant. 2020 Nov;55(11):2071-2076. doi: 10.1038/s41409-020-0919-0. Epub 2020 May 13. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

