Orientational Jahn-Teller Isomerism in the Dark-Stable State of Nature's Water Oxidase
- PMID: 33830630
- PMCID: PMC8252073
- DOI: 10.1002/anie.202103425
Orientational Jahn-Teller Isomerism in the Dark-Stable State of Nature's Water Oxidase
Abstract
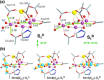
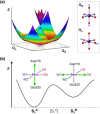
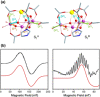
The tetramanganese-calcium cluster of the oxygen-evolving complex of photosystem II adopts electronically and magnetically distinct but interconvertible valence isomeric forms in its first light-driven oxidized catalytic state, S2 . This bistability is implicated in gating the final catalytic states preceding O-O bond formation, but it is unknown how the biological system enables its emergence and controls its effect. Here we show that the Mn4 CaO5 cluster in the resting (dark-stable) S1 state adopts orientational Jahn-Teller isomeric forms arising from a directional change in electronic configuration of the "dangler" MnIII ion. The isomers are consistent with available structural data and explain previously unresolved electron paramagnetic resonance spectroscopic observations on the S1 state. This unique isomerism in the resting state is shown to be the electronic origin of valence isomerism in the S2 state, establishing a functional role of orientational Jahn-Teller isomerism unprecedented in biological or artificial catalysis.
Keywords: EPR spectroscopy; bioinorganic chemistry; computational chemistry; electronic structure; photosynthesis.
© 2021 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

