The Hsp70 chaperone system: distinct roles in erythrocyte formation and maintenance
- PMID: 33832207
- PMCID: PMC8168490
- DOI: 10.3324/haematol.2019.233056
The Hsp70 chaperone system: distinct roles in erythrocyte formation and maintenance
Abstract
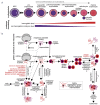
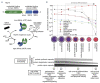
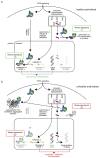
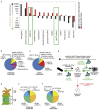
Erythropoiesis is a tightly regulated cell differentiation process in which specialized oxygen- and carbon dioxide-carrying red blood cells are generated in vertebrates. Extensive reorganization and depletion of the erythroblast proteome leading to the deterioration of general cellular protein quality control pathways and rapid hemoglobin biogenesis rates could generate misfolded/aggregated proteins and trigger proteotoxic stresses during erythropoiesis. Such cytotoxic conditions could prevent proper cell differentiation resulting in premature apoptosis of erythroblasts (ineffective erythropoiesis). The heat shock protein 70 (Hsp70) molecular chaperone system supports a plethora of functions that help maintain cellular protein homeostasis (proteostasis) and promote red blood cell differentiation and survival. Recent findings show that abnormalities in the expression, localization and function of the members of this chaperone system are linked to ineffective erythropoiesis in multiple hematological diseases in humans. In this review, we present latest advances in our understanding of the distinct functions of this chaperone system in differentiating erythroblasts and terminally differentiated mature erythrocytes. We present new insights into the protein repair-only function(s) of the Hsp70 system, perhaps to minimize protein degradation in mature erythrocytes to warrant their optimal function and survival in the vasculature under healthy conditions. The work also discusses the modulatory roles of this chaperone system in a wide range of hematological diseases and the therapeutic gain of targeting Hsp70.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

