Repurposing cabozantinib with therapeutic potential in KIT-driven t(8;21) acute myeloid leukaemias
- PMID: 33833412
- PMCID: PMC9113930
- DOI: 10.1038/s41417-021-00329-1
Repurposing cabozantinib with therapeutic potential in KIT-driven t(8;21) acute myeloid leukaemias
Abstract
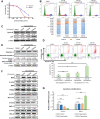
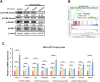
Cabozantinib is an orally available, multi-target tyrosine kinase inhibitor approved for the treatment of several solid tumours and known to inhibit KIT tyrosine kinase. In acute myeloid leukaemia (AML), aberrant KIT tyrosine kinase often coexists with t(8;21) to drive leukaemogenesis. Here we evaluated the potential therapeutic effect of cabozantinib on a selected AML subtype characterised by t(8;21) coupled with KIT mutation. Cabozantinib exerted substantial cytotoxicity in Kasumi-1 cells with an IC50 of 88.06 ± 4.32 nM, which was well within clinically achievable plasma levels. The suppression of KIT phosphorylation and its downstream signals, including AKT/mTOR, STAT3, and ERK1/2, was elicited by cabozantinib treatment and associated with subsequent alterations of cell cycle- and apoptosis-related molecules. Cabozantinib also disrupted the synthesis of an AML1-ETO fusion protein in a dose- and time-dependent manner. In a mouse xenograft model, cabozantinib suppressed tumourigenesis at 10 mg/kg and significantly prolonged survival of the mice. Further RNA-sequencing analysis revealed that mTOR-mediated signalling pathways were substantially inactivated by cabozantinib treatment, causing the downregulation of ribosome biogenesis and glycolysis, along with myeloid leukocyte activation. We suggest that cabozantinib may be effective in the treatment of AML with t(8;21) and KIT mutation. Relevant clinical trials are warranted.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

