Conditional RAC1 knockout in motor neurons restores H-reflex rate-dependent depression after spinal cord injury
- PMID: 33837249
- PMCID: PMC8035187
- DOI: 10.1038/s41598-021-87476-5
Conditional RAC1 knockout in motor neurons restores H-reflex rate-dependent depression after spinal cord injury
Abstract
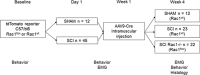
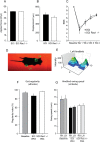
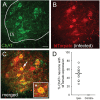
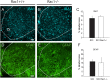
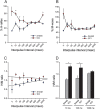
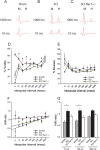
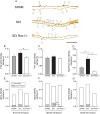
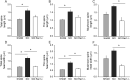
A major complication with spinal cord injury (SCI) is the development of spasticity, a clinical symptom of hyperexcitability within the spinal H-reflex pathway. We have previously demonstrated a common structural motif of dendritic spine dysgenesis associated with hyperexcitability disorders after injury or disease insults to the CNS. Here, we used an adeno-associated viral (AAV)-mediated Cre-Lox system to knockout Rac1 protein expression in motor neurons after SCI. Three weeks after AAV9-Cre delivery into the soleus/gastrocnemius of Rac1-"floxed" adult mice to retrogradely infect spinal alpha-motor neurons, we observed significant restoration of RDD and reduced H-reflex excitability in SCI animals. Additionally, viral-mediated Rac1 knockdown reduced presence of dendritic spine dysgenesis on motor neurons. In control SCI animals without Rac1 knockout, we continued to observe abnormal dendritic spine morphology associated with hyperexcitability disorder, including an increase in mature, mushroom dendritic spines, and an increase in overall spine length and spine head size. Taken together, our results demonstrate that viral-mediated disruption of Rac1 expression in ventral horn motor neurons can mitigate dendritic spine morphological correlates of neuronal hyperexcitability, and reverse hyperreflexia associated with spasticity after SCI. Finally, our findings provide evidence of a putative mechanistic relationship between motor neuron dendritic spine dysgenesis and SCI-induced spasticity.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Conditional Astrocyte Rac1KO Attenuates Hyperreflexia after Spinal Cord Injury.J Neurosci. 2024 Jan 3;44(1):e1670222023. doi: 10.1523/JNEUROSCI.1670-22.2023. J Neurosci. 2024. PMID: 37963762 Free PMC article.
-
PAK1 inhibition with Romidepsin attenuates H-reflex hyperexcitability after spinal cord injury.J Physiol. 2024 Oct;602(19):5061-5081. doi: 10.1113/JP284976. Epub 2024 Sep 4. J Physiol. 2024. PMID: 39231098
-
Dendritic spine dysgenesis contributes to hyperreflexia after spinal cord injury.J Neurophysiol. 2015 Mar 1;113(5):1598-615. doi: 10.1152/jn.00566.2014. Epub 2014 Dec 10. J Neurophysiol. 2015. PMID: 25505110 Free PMC article.
-
Dendritic spine dysgenesis in neuropathic pain.Neurosci Lett. 2015 Aug 5;601:54-60. doi: 10.1016/j.neulet.2014.11.024. Epub 2014 Nov 20. Neurosci Lett. 2015. PMID: 25445354 Review.
-
Dendritic spine dysgenesis in neuropathic pain.Prog Mol Biol Transl Sci. 2015;131:385-408. doi: 10.1016/bs.pmbts.2014.12.001. Epub 2015 Feb 7. Prog Mol Biol Transl Sci. 2015. PMID: 25744680 Review.
Cited by
-
Increased astrocytic GLT-1 expression in tripartite synapses is associated with SCI-induced hyperreflexia.J Neurophysiol. 2023 Nov 1;130(5):1358-1366. doi: 10.1152/jn.00234.2023. Epub 2023 Oct 25. J Neurophysiol. 2023. PMID: 37877184 Free PMC article.
-
Molecular Identification of Pro-Excitogenic Receptor and Channel Phenotypes of the Deafferented Lumbar Motoneurons in the Early Phase after SCT in Rats.Int J Mol Sci. 2022 Sep 22;23(19):11133. doi: 10.3390/ijms231911133. Int J Mol Sci. 2022. PMID: 36232433 Free PMC article.
-
Conditional Astrocyte Rac1KO Attenuates Hyperreflexia after Spinal Cord Injury.J Neurosci. 2024 Jan 3;44(1):e1670222023. doi: 10.1523/JNEUROSCI.1670-22.2023. J Neurosci. 2024. PMID: 37963762 Free PMC article.
-
DOCK3-Associated Neurodevelopmental Disorder-Clinical Features and Molecular Basis.Genes (Basel). 2023 Oct 14;14(10):1940. doi: 10.3390/genes14101940. Genes (Basel). 2023. PMID: 37895289 Free PMC article. Review.
-
Rate-Dependent Depression of the Hoffmann Reflex: Practical Applications in Painful Diabetic Neuropathy.Diabetes Metab J. 2024 Nov;48(6):1029-1046. doi: 10.4093/dmj.2024.0614. Epub 2024 Nov 21. Diabetes Metab J. 2024. PMID: 39610132 Free PMC article. Review.
References
-
- Walter JS, et al. A database of self-reported secondary medical problems among VA spinal cord injury patients: its role in clinical care and management. J. Rehabil. Res. Dev. 2002;39:53–61. - PubMed
-
- Lance JW. In: Spasticity: Disordered Motor Control. Feldman RG, Yound RR, Koella WP, editors. Year Book Medical; 1980. pp. 485–494.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

