RB1CC1 functions as a tumor-suppressing gene in renal cell carcinoma via suppression of PYK2 activity and disruption of TAZ-mediated PDL1 transcription activation
- PMID: 33837850
- PMCID: PMC10992581
- DOI: 10.1007/s00262-021-02913-8
RB1CC1 functions as a tumor-suppressing gene in renal cell carcinoma via suppression of PYK2 activity and disruption of TAZ-mediated PDL1 transcription activation
Abstract
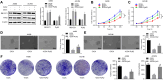
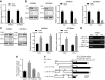
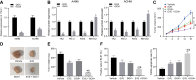
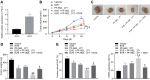
Rb1-inducible coiled-coil 1 (RB1CC1) has been demonstrated to function as an inhibitor of proline-rich/Ca-activated tyrosine kinase 2 (PYK2) by binding to the kinase domain of PYK2, which promotes the proliferation, invasion, and migration of renal cell carcinoma (RCC) cells. Additionally, in breast cancer, PYK2 positively regulates the expression of transcriptional co-activator with PDZ-binding motif (TAZ) which in turn can enhance PDL1 levels in breast and lung cancer cells. The current study was performed to decipher the impact of RB1CC1 in the progression of RCC via regulation of the PYK2/TAZ/PDL1 signaling axis. Expression of RB1CC1 and PYK2 was quantified in clinical tissue samples from RCC patients. The relationship between TAZ and PYK2, TAZ and PDL1 was then validated. The cellular processes of doxorubicin (DOX)-induced human RCC cell lines including the abilities of proliferation, colony formation, sphere formation and apoptosis, as well as the tumorigenicity of transfected cells, were evaluated after the alteration of RB1CC1 expression. RB1CC1 exhibited decreased expression in RCC tissues and was positively correlated with patient survival. RB1CC1 could inhibit the activity of PYK2, which in turn stimulated the stability of TAZ protein by phosphorylating TAZ. Meanwhile, TAZ protein activated PDL1 transcription by binding to the promoter region of PDL1. RB1CC1 overexpression or PYK2 knockdown could help everolimus (EVE) to inhibit tumor proliferation and activate immune response. Taken together, RB1CC1 can potentially augment the response of RCC cells to immunotherapy by suppressing the PYK2/TAZ/PDL1 signaling axis.
Keywords: Programmed cell death ligand 1; Proline-rich/Ca-activated tyrosine kinase 2; Rb1-inducible coiled-coil 1; Renal cell carcinoma; Tafazzin.
© 2021. The Author(s), under exclusive licence to Springer-Verlag GmbH Germany, part of Springer Nature.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures




Similar articles
-
PYK2 negatively regulates the Hippo pathway in TNBC by stabilizing TAZ protein.Cell Death Dis. 2018 Sep 24;9(10):985. doi: 10.1038/s41419-018-1005-z. Cell Death Dis. 2018. PMID: 30250159 Free PMC article.
-
Programmed Death Ligand 1; An Immunotarget for Renal Cell Carcinoma.Asian Pac J Cancer Prev. 2019 Oct 1;20(10):2951-2957. doi: 10.31557/APJCP.2019.20.10.2951. Asian Pac J Cancer Prev. 2019. PMID: 31653140 Free PMC article.
-
Centromere protein K enhances the activation of YAP1/TAZ signal cascade to drive the progression of clear cell renal cell carcinoma.Toxicol Appl Pharmacol. 2025 Jan;494:117181. doi: 10.1016/j.taap.2024.117181. Epub 2024 Nov 29. Toxicol Appl Pharmacol. 2025. PMID: 39617260
-
The Hippo Pathway Effector TAZ Regulates Ferroptosis in Renal Cell Carcinoma.Cell Rep. 2019 Sep 3;28(10):2501-2508.e4. doi: 10.1016/j.celrep.2019.07.107. Cell Rep. 2019. PMID: 31484063 Free PMC article.
-
The interaction of YBX1 with G3BP1 promotes renal cell carcinoma cell metastasis via YBX1/G3BP1-SPP1- NF-κB signaling axis.J Exp Clin Cancer Res. 2019 Sep 3;38(1):386. doi: 10.1186/s13046-019-1347-0. J Exp Clin Cancer Res. 2019. PMID: 31481087 Free PMC article.
Cited by
-
Reduced Proline-Rich Tyrosine Kinase 2 Promotes Tumor Metastasis by Activating Epithelial-Mesenchymal Transition in Colorectal Cancer.Dig Dis Sci. 2024 Nov;69(11):4098-4107. doi: 10.1007/s10620-024-08643-y. Epub 2024 Oct 16. Dig Dis Sci. 2024. PMID: 39414740
-
In silico analysis and experimental validation shows negative correlation between miR-1183 and cell cycle progression gene 1 expression in colorectal cancer.PLoS One. 2023 Aug 4;18(8):e0289082. doi: 10.1371/journal.pone.0289082. eCollection 2023. PLoS One. 2023. PMID: 37540697 Free PMC article.
-
Emerging Role of Hippo-YAP (Yes-Associated Protein)/TAZ (Transcriptional Coactivator with PDZ-Binding Motif) Pathway Dysregulation in Renal Cell Carcinoma Progression.Cancers (Basel). 2024 Aug 3;16(15):2758. doi: 10.3390/cancers16152758. Cancers (Basel). 2024. PMID: 39123485 Free PMC article. Review.
-
METTL14 inhibits malignant progression of oral squamous cell carcinoma by targeting the autophagy-related gene RB1CC1 in an m6A-IGF2BP2-dependent manner.Clin Sci (Lond). 2023 Sep 13;137(17):1373-1389. doi: 10.1042/CS20230219. Clin Sci (Lond). 2023. PMID: 37615536 Free PMC article.
-
Frontier knowledge and future directions of programmed cell death in clear cell renal cell carcinoma.Cell Death Discov. 2024 Mar 5;10(1):113. doi: 10.1038/s41420-024-01880-0. Cell Death Discov. 2024. PMID: 38443363 Free PMC article. Review.
References
-
- Billon E, Walz J, Brunelle S, Thomassin J, Salem N, Guerin M, Vicier C, Dermeche S, Albiges L, Tantot F, Nenan S, Pignot G, Gravis G. Vitiligo adverse event observed in a patient with durable complete response after nivolumab for metastatic renal cell carcinoma. Front Oncol. 2019;9:1033. doi: 10.3389/fonc.2019.01033. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

