Early Life Microbiota Colonization at Six Months of Age: A Transitional Time Point
- PMID: 33842380
- PMCID: PMC8032992
- DOI: 10.3389/fcimb.2021.590202
Early Life Microbiota Colonization at Six Months of Age: A Transitional Time Point
Abstract
Background: Early life gut microbiota is involved in several biological processes, particularly metabolism, immunity, and cognitive neurodevelopment. Perturbation in the infant's gut microbiota increases the risk for diseases in early and later life, highlighting the importance of understanding the connections between perinatal factors with early life microbial composition. The present research paper is aimed at exploring the prenatal and postnatal factors influencing the infant gut microbiota composition at six months of age.
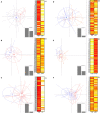
Methods: Gut microbiota of infants enrolled in the longitudinal, prospective, observational study "A.MA.MI" (Alimentazione MAmma e bambino nei primi MIlle giorni) was analyzed. We collected and analyzed 61 fecal samples at baseline (meconium, T0); at six months of age (T2), we collected and analyzed 53 fecal samples. Samples were grouped based on maternal and gestational weight factors, type of delivery, type of feeding, time of weaning, and presence/absence of older siblings. Alpha and beta diversities were evaluated to describe microbiota composition. Multivariate analyses were performed to understand the impact of the aforementioned factors on the infant's microbiota composition at six months of age.
Results: Different clustering hypotheses have been tested to evaluate the impact of known metadata factors on the infant microbiota. Neither maternal body mass index nor gestational weight gain was able to determine significant differences in infant microbiota composition six months of age. Concerning the type of feeding, we observed a low alpha diversity in exclusive breastfed infants; conversely, non-exclusively breastfed infants reported an overgrowth of Ruminococcaceae and Flavonifractor. Furthermore, we did not find any statistically significant difference resulting from an early introduction of solid foods (before 4 months of age). Lastly, our sample showed a higher abundance of clostridial patterns in firstborn babies when compared to infants with older siblings in the family.
Conclusion: Our findings showed that, at this stage of life, there is not a single factor able to affect in a distinct way the infants' gut microbiota development. Rather, there seems to be a complex multifactorial interaction between maternal and neonatal factors determining a unique microbial niche in the gastrointestinal tract.
Keywords: breast milk; delivery; maternal factors; neonatal microbiota; newborn; older siblings; weaning.
Copyright © 2021 Raspini, Vacca, Porri, De Giuseppe, Calabrese, Chieppa, Liso, Cerbo, Civardi, Garofoli, De Angelis and Cena.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
-
- Azad M. B., Konya T., Maughan H., Guttman D. S., Field C. J., Sears M. R., et al. (2013). Infant gut microbiota and the hygiene hypothesis of allergic disease: impact of household pets and siblings on microbiota composition and diversity. Allergy Asthma Clin. Immunol. 9 (1), 15. 10.1186/1710-1492-9-15 - DOI - PMC - PubMed
-
- Biddle A., Stewart L., Blanchard J., Leschine S. (2013). Untangling the genetic basis of fibrolytic specialization by Lachnospiraceae and Ruminococcaceae in diverse gut communities. Diversity 5 (3), 627–640. 10.3390/d5030627 - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

