Dermatomyositis autoantibodies: how can we maximize utility?
- PMID: 33842654
- PMCID: PMC8033377
- DOI: 10.21037/atm-20-5175
Dermatomyositis autoantibodies: how can we maximize utility?
Abstract
The past 15 years has seen significant advances in the characterization of myositis-specific autoantibodies (MSAs) and their associated phenotypes in patients with dermatomyositis (DM). As more careful studies are performed, it is clear that unique combinations of clinical and pathological phenotypes are associated with each MSA, despite the fact that there is considerable heterogeneity within antibody classes as well as overlap across the groups. Because risk for interstitial lung disease (ILD), internal malignancy, adverse disease trajectory, and, potentially response to therapy differ by DM MSA group, a deeper understanding of MSAs and validation and standardization of assays used for detection are critical for optimizing diagnosis and treatment. Like any test, the diagnostic sensitivity and specificity of assays for various MSAs is not perfect. Currently tests for MSAs are helpful at minimum for a clinician to assess relative risk or contribute to diagnosis and perhaps counsel the appropriate patient about what to expect. With international standardization and larger studies it is likely that more antibody tests will make their way into formal schemata for diagnosis and actionable risk assessment in DM. In this review, we summarize key considerations for interpreting the clinical and pathologic associations with MSA in DM and identify critical gaps in knowledge and practice that will maximize their clinical utility and utility for understanding disease pathogenesis.
Keywords: Dermatomyositis (DM); autoantibodies; immunoassay.
2021 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-5175). The series “Rheumatologic Skin Disease” was commissioned by the editorial office without any funding or sponsorship. DFF served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
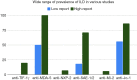
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
