Ferritinophagy is involved in the zinc oxide nanoparticles-induced ferroptosis of vascular endothelial cells
- PMID: 33843441
- PMCID: PMC8726675
- DOI: 10.1080/15548627.2021.1911016
Ferritinophagy is involved in the zinc oxide nanoparticles-induced ferroptosis of vascular endothelial cells
Abstract
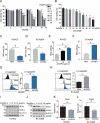
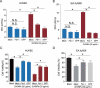
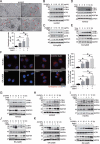
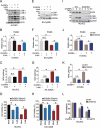
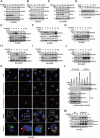
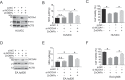
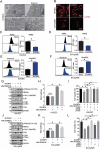
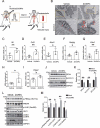
Zinc oxide nanoparticles (ZnONPs) hold great promise for biomedical applications. Previous studies have revealed that ZnONPs exposure can induce toxicity in endothelial cells, but the underlying mechanisms have not been fully elucidated. In this study, we report that ZnONPs can induce ferroptosis of both HUVECs and EA.hy926 cells, as evidenced by the elevation of intracellular iron levels, lipid peroxidation and cell death in a dose- and time-dependent manner. In addition, both the lipid reactive oxygen species (ROS) scavenger ferrostatin-1 and the iron chelator deferiprone attenuated ZnONPs-induced cell death. Intriguingly, we found that ZnONPs-induced ferroptosis is macroautophagy/autophagy-dependent, because the inhibition of autophagy with a pharmacological inhibitor or by ATG5 gene knockout profoundly mitigated ZnONPs-induced ferroptosis. We further demonstrated that NCOA4 (nuclear receptor coactivator 4)-mediated ferritinophagy (autophagic degradation of the major intracellular iron storage protein ferritin) was required for the ferroptosis induced by ZnONPs, by showing that NCOA4 knockdown can reduce the intracellular iron level and lipid peroxidation, and subsequently alleviate ZnONPs-induced cell death. Furthermore, we showed that ROS originating from mitochondria (mtROS) probably activated the AMPK-ULK1 axis to trigger ferritinophagy. Most importantly, pulmonary ZnONPs exposure caused vascular inflammation and ferritinophagy in mice, and ferrostatin-1 supplementation significantly reversed the vascular injury induced by pulmonary ZnONPs exposure. Overall, our study indicates that ferroptosis is a novel mechanism for ZnONPs-induced endothelial cytotoxicity, and that NCOA4-mediated ferritinophagy is required for ZnONPs-induced ferroptotic cell death.Abbreviations: 3-MA: 3-methyladenine; ACTB: Actin beta; AMPK: AMP-activated protein kinase; ATG: Autophagy-related; BafA1: Bafilomycin A1; CQ: Choloroquine; DFP: Deferiprone; FACS: Fluorescence-activated cell sorting; Fer-1: Ferrostatin-1; FTH1: Ferritin heavy chain 1; GPX4: Glutathione peroxidase 4; GSH: Glutathione; IREB2/IRP2: Iron responsive element binding protein 2; LIP: Labile iron pool; MAP1LC3B/LC3B: Microtubule associated protein 1 light chain 3 beta; MTOR: Mechanistic target of rapamycin kinase; NCOA4: Nuclear receptor coactivator 4; NFE2L2/NRF2: Nuclear factor, erythroid 2 like 2; PGSK: Phen Green™ SK; ROS: Reactive oxygen species; siRNA: Small interfering RNA; SQSTM1/p62: Sequestosome 1; TEM: Transmission electron microscopy; ULK1: Unc-51 like autophagy activating kinase 1; ZnONPs: Zinc oxide nanoparticles.
Keywords: Autophagy; ferritinophagy; ferroptosis; vascular endothelial cell; zinc oxide nanoparticles.
Conflict of interest statement
The authors declare that they have no competing financial interests.
Figures


Similar articles
-
Activation of ferritinophagy is required for the RNA-binding protein ELAVL1/HuR to regulate ferroptosis in hepatic stellate cells.Autophagy. 2018;14(12):2083-2103. doi: 10.1080/15548627.2018.1503146. Epub 2018 Aug 21. Autophagy. 2018. PMID: 30081711 Free PMC article.
-
ATM orchestrates ferritinophagy and ferroptosis by phosphorylating NCOA4.Autophagy. 2023 Jul;19(7):2062-2077. doi: 10.1080/15548627.2023.2170960. Epub 2023 Feb 8. Autophagy. 2023. PMID: 36752571 Free PMC article.
-
STAT3 signaling promotes cardiac injury by upregulating NCOA4-mediated ferritinophagy and ferroptosis in high-fat-diet fed mice.Free Radic Biol Med. 2023 May 20;201:111-125. doi: 10.1016/j.freeradbiomed.2023.03.003. Epub 2023 Mar 20. Free Radic Biol Med. 2023. PMID: 36940731
-
Lipid Peroxidation and Iron Metabolism: Two Corner Stones in the Homeostasis Control of Ferroptosis.Int J Mol Sci. 2022 Dec 27;24(1):449. doi: 10.3390/ijms24010449. Int J Mol Sci. 2022. PMID: 36613888 Free PMC article. Review.
-
The Role of NCOA4-Mediated Ferritinophagy in Ferroptosis.Adv Exp Med Biol. 2021;1301:41-57. doi: 10.1007/978-3-030-62026-4_4. Adv Exp Med Biol. 2021. PMID: 34370287 Review.
Cited by
-
Hepcidin depending on astrocytic NEO1 ameliorates blood-brain barrier dysfunction after subarachnoid hemorrhage.Cell Death Dis. 2024 Aug 7;15(8):569. doi: 10.1038/s41419-024-06909-x. Cell Death Dis. 2024. PMID: 39107268 Free PMC article.
-
Iron metabolism, ferroptosis, and lncRNA in cancer: knowns and unknowns.J Zhejiang Univ Sci B. 2022 Oct 15;23(10):844-862. doi: 10.1631/jzus.B2200194. J Zhejiang Univ Sci B. 2022. PMID: 36226538 Free PMC article. Review.
-
Autophagy-mediated ferroptosis is involved in development of severe acute pancreatitis.BMC Gastroenterol. 2024 Aug 1;24(1):245. doi: 10.1186/s12876-024-03345-1. BMC Gastroenterol. 2024. PMID: 39090535 Free PMC article.
-
Exploring Dysregulated Ferroptosis-Related Genes in Septic Myocardial Injury Based on Human Heart Transcriptomes: Evidence and New Insights.J Inflamm Res. 2023 Mar 9;16:995-1015. doi: 10.2147/JIR.S400107. eCollection 2023. J Inflamm Res. 2023. PMID: 36923465 Free PMC article.
-
Deferiprone: A Forty-Year-Old Multi-Targeting Drug with Possible Activity against COVID-19 and Diseases of Similar Symptomatology.Int J Mol Sci. 2022 Jun 16;23(12):6735. doi: 10.3390/ijms23126735. Int J Mol Sci. 2022. PMID: 35743183 Free PMC article. Review.
References
-
- Xiong HM. ZnO nanoparticles applied to bioimaging and drug delivery. Adv Mater. 2013;25(37):5329–5335. - PubMed
-
- Kao YY, Chen YC, Cheng TJ, et al. Zinc oxide nanoparticles interfere with zinc ion homeostasis to cause cytotoxicity. Toxicol Sci. 2012;125(2):462–472. - PubMed
-
- Heim J, Felder E, Tahir MN, et al. Genotoxic effects of zinc oxide nanoparticles. Nanoscale. 2015;7(19):8931–8938. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
