ATF3 deficiency impairs the proliferative-secretory phase transition and decidualization in RIF patients
- PMID: 33846304
- PMCID: PMC8041902
- DOI: 10.1038/s41419-021-03679-8
ATF3 deficiency impairs the proliferative-secretory phase transition and decidualization in RIF patients
Abstract
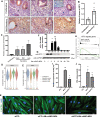
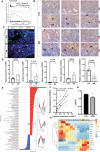
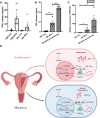
Decidualization is a complex process involving cellular proliferation and differentiation of the endometrial stroma and is required to establish and support pregnancy. Dysregulated decidualization has been reported to be a critical cause of recurrent implantation failure (RIF). In this study, we found that Activating transcription factor 3 (ATF3) expression was significantly downregulated in the endometrium of RIF patients. Knockdown of ATF3 in human endometrium stromal cells (hESCs) hampers decidualization, while overexpression could trigger the expression of decidual marker genes, and ameliorate the decidualization of hESCs from RIF patients. Mechanistically, ATF3 promotes decidualization by upregulating FOXO1 via suppressing miR-135b expression. In addition, the endometrium of RIF patients was hyperproliferative, while overexpression of ATF3 inhibited the proliferation of hESCs through CDKN1A. These data demonstrate the critical roles of endometrial ATF3 in regulating decidualization and proliferation, and dysregulation of ATF3 in the endometrium may be a novel cause of RIF and therefore represent a potential therapeutic target for RIF.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
A novel lncRNA lncSAMD11-1: 1 interacts with PIP4K2A to promote endometrial decidualization by stabilizing FoxO1 nuclear localization.Int J Biochem Cell Biol. 2022 Oct;151:106280. doi: 10.1016/j.biocel.2022.106280. Epub 2022 Aug 17. Int J Biochem Cell Biol. 2022. PMID: 35987479
-
Increased Krüppel-like factor 12 in recurrent implantation failure impairs endometrial decidualization by repressing Nur77 expression.Reprod Biol Endocrinol. 2017 Mar 31;15(1):25. doi: 10.1186/s12958-017-0243-8. Reprod Biol Endocrinol. 2017. PMID: 28359310 Free PMC article.
-
Decreased CD44v3 expression impairs endometrial stromal cell proliferation and decidualization in women with recurrent implantation failure.Reprod Biol Endocrinol. 2022 Dec 16;20(1):170. doi: 10.1186/s12958-022-01042-w. Reprod Biol Endocrinol. 2022. PMID: 36527033 Free PMC article.
-
FoxO1 is a cell-specific core transcription factor for endometrial remodeling and homeostasis during menstrual cycle and early pregnancy.Hum Reprod Update. 2021 Apr 21;27(3):570-583. doi: 10.1093/humupd/dmaa060. Hum Reprod Update. 2021. PMID: 33434267 Review.
-
Glucose metabolism and endometrium decidualization.Front Endocrinol (Lausanne). 2025 Feb 17;16:1546335. doi: 10.3389/fendo.2025.1546335. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40034230 Free PMC article. Review.
Cited by
-
Molecular Clues to Understanding Causes of Human-Assisted Reproduction Treatment Failures and Possible Treatment Options.Int J Mol Sci. 2022 Sep 8;23(18):10357. doi: 10.3390/ijms231810357. Int J Mol Sci. 2022. PMID: 36142268 Free PMC article. Review.
-
Exploring the role of Prx II in mitigating endoplasmic reticulum stress and mitochondrial dysfunction in neurodegeneration.Cell Commun Signal. 2024 Apr 18;22(1):231. doi: 10.1186/s12964-024-01613-x. Cell Commun Signal. 2024. PMID: 38637880 Free PMC article.
-
Kaempferol, a Phytoprogestin, Induces a Subset of Progesterone-Regulated Genes in the Uterus.Nutrients. 2023 Mar 15;15(6):1407. doi: 10.3390/nu15061407. Nutrients. 2023. PMID: 36986136 Free PMC article.
-
Dysregulation of endometrial stromal serotonin homeostasis leading to abnormal phosphatidylcholine metabolism impairs decidualization in patients with recurrent implantation failure.Hum Reprod Open. 2024 Jun 20;2024(3):hoae042. doi: 10.1093/hropen/hoae042. eCollection 2024. Hum Reprod Open. 2024. PMID: 39091587 Free PMC article.
-
Downregulated INHBB in endometrial tissue of recurrent implantation failure patients impeded decidualization through the ADCY1/cAMP signalling pathway.J Assist Reprod Genet. 2023 May;40(5):1135-1146. doi: 10.1007/s10815-023-02762-7. Epub 2023 Mar 13. J Assist Reprod Genet. 2023. PMID: 36913138 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

