The Use, Standardization, and Interpretation of Brain Imaging Data in Clinical Trials of Neurodegenerative Disorders
- PMID: 33846962
- PMCID: PMC8423963
- DOI: 10.1007/s13311-021-01027-4
The Use, Standardization, and Interpretation of Brain Imaging Data in Clinical Trials of Neurodegenerative Disorders
Erratum in
-
Correction to: The Use, Standardization, and Interpretation of Brain Imaging Data in Clinical Trials of Neurodegenerative Disorders.Neurotherapeutics. 2021 Jul;18(3):2127. doi: 10.1007/s13311-021-01063-0. Neurotherapeutics. 2021. PMID: 33904114 Free PMC article. No abstract available.
Abstract
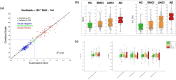
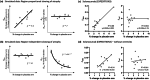
Imaging biomarkers play a wide-ranging role in clinical trials for neurological disorders. This includes selecting the appropriate trial participants, establishing target engagement and mechanism-related pharmacodynamic effect, monitoring safety, and providing evidence of disease modification. In the early stages of clinical drug development, evidence of target engagement and/or downstream pharmacodynamic effect-especially with a clear relationship to dose-can provide confidence that the therapeutic candidate should be advanced to larger and more expensive trials, and can inform the selection of the dose(s) to be further tested, i.e., to "de-risk" the drug development program. In these later-phase trials, evidence that the therapeutic candidate is altering disease-related biomarkers can provide important evidence that the clinical benefit of the compound (if observed) is grounded in meaningful biological changes. The interpretation of disease-related imaging markers, and comparability across different trials and imaging tools, is greatly improved when standardized outcome measures are defined. This standardization should not impinge on scientific advances in the imaging tools per se but provides a common language in which the results generated by these tools are expressed. PET markers of pathological protein aggregates and structural imaging of brain atrophy are common disease-related elements across many neurological disorders. However, PET tracers for pathologies beyond amyloid β and tau are needed, and the interpretability of structural imaging can be enhanced by some simple considerations to guard against the possible confound of pseudo-atrophy. Learnings from much-studied conditions such as Alzheimer's disease and multiple sclerosis will be beneficial as the field embraces rarer diseases.
Keywords: Clinical trials; Imaging; MRI; Neurology; PET.
© 2021. The Author(s).
Figures




References
-
- Group, F.-N.B.W., BEST (Biomarkers, EndpointS, and other Tools) Resource . Food and Drug Administration (US) & National Institutes of Health (US): Silver Spring. MD: MD & Bethesda; 2016. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

