Which is the most effective one in knee osteoarthritis treatment from mesenchymal stem cells obtained from different sources?-A systematic review with conventional and network meta-analyses of randomized controlled trials
- PMID: 33850849
- PMCID: PMC8039709
- DOI: 10.21037/atm-20-5116
Which is the most effective one in knee osteoarthritis treatment from mesenchymal stem cells obtained from different sources?-A systematic review with conventional and network meta-analyses of randomized controlled trials
Abstract
Background: Due to varying degrees of difficulty in obtaining different mesenchymal stem cells (MSCs), the distinct pain levels and treatment costs, and for providing concrete evidence for future clinical practice, a thorough comparison of all relevant MSCs remained critical. Hence, this study aimed to achieve this objective to compare the efficacy of MSCs obtained from different sources in clinical outcomes and cartilage repair of knee osteoarthritis (KOA).
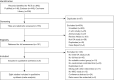
Methods: The EmBase, PubMed and Cochrane Library databases were searched for eligible studies. Randomized controlled trials (RCTs) that compared MSCs from different sources with placebo or each other in KOA patients. Conventional meta-analysis and frequentist network meta-analysis (NMA) were conducted. The primary clinical outcome was pain relief. The frequentist NMA was conducted using Stata with the "network" command.
Results: Eight studies (seven trials) involving 203 KOA patients were included in this meta-analysis. The MSCs were considered superior over placebo for pain relief and improved function in KOA, but showed no statistically significant differences for cartilage regeneration. Among all the MSCs, the adipose tissue-derived MSCs (AD-MSCs) most effectively relieved pain.
Conclusion: These findings suggested that MSCs are effective in the treating of KOA. AD-MSCs might be the most effective for relieving pain, and Umbilical cord-derived mesenchymal stem cells (UC-MSCs) might be the most effective for improving function. However, the current evidence does not support the use of MSCs for improving cartilage repair in KOA patients.
Keywords: Relative efficacy; knee osteoarthritis (KOA); mesenchymal stem cells (MSCs); meta-analyses; randomized controlled trials.
2021 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-20-5116). The authors have no conflicts of interest to declare.
Figures




References
LinkOut - more resources
Full Text Sources
Other Literature Sources
