Cellular responses and functions of α7 nicotinic acetylcholine receptor activation in the brain: a narrative review
- PMID: 33850906
- PMCID: PMC8039675
- DOI: 10.21037/atm-21-273
Cellular responses and functions of α7 nicotinic acetylcholine receptor activation in the brain: a narrative review
Abstract
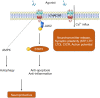
The α7 nicotinic acetylcholine receptor (α7nAChR) has been studied for many years since its discovery. Although many functions and characteristics of brain α7nAChR are widely understood, much remains to be elucidated. The α7nAChR is widely expressed in the central nervous system, not only in neurons but also in astrocytes, microglia, and endothelial cells. α7nAChR can be activated by endogenous agonist like acetylcholine or exogenous agonists like nicotine and PNU282987. Its agonists can be divided into selective agonists and non-selective agonists. The activation of α7nAChR results in a series of physiological processes which have both short-term and long-term effects on cells, for example, calcium influx, neurotransmitter release, synaptic plasticity, and excitatory transmission. It also induces other downstream events, such as inflammation, autophagy, necrosis, transcription, and apoptosis. The cellular responses to α7nAChR activation vary according to cell types and conditions. For example, α7nAChR activation in pyramidal neurons leads to long-term potentiation, while α7nAChR activation in GABAergic interneurons leads to long-term depression. Studies have also shown some contradictory phenomena, which requires further study for clarification. Herein, the cellular responses of α7nAChR activation are summarized, and the functions of α7nAChR in neurons and non-neuronal cells are discussed. We also summarized contradictory conclusions to show where we stand and where to go for future studies.
Keywords: calcium influx; central nervous system diseases (CNS diseases); signaling pathway; synaptic plasticity; α7 nicotinic acetylcholine receptor (α7nAChR).
2021 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-21-273). The authors have no conflicts of interest to declare.
Figures
Similar articles
-
The p38 mitogen activated protein kinase regulates β-amyloid protein internalization through the α7 nicotinic acetylcholine receptor in mouse brain.Brain Res Bull. 2018 Mar;137:41-52. doi: 10.1016/j.brainresbull.2017.11.006. Epub 2017 Nov 9. Brain Res Bull. 2018. PMID: 29128415
-
Targeting the nicotinic alpha7 acetylcholine receptor to enhance cognition in disease.Biochem Pharmacol. 2011 Oct 15;82(8):891-903. doi: 10.1016/j.bcp.2011.06.034. Epub 2011 Jul 2. Biochem Pharmacol. 2011. PMID: 21741954 Review.
-
α7 Nicotinic Acetylcholine Receptor Signaling Modulates Ovine Fetal Brain Astrocytes Transcriptome in Response to Endotoxin.Front Immunol. 2019 May 9;10:1063. doi: 10.3389/fimmu.2019.01063. eCollection 2019. Front Immunol. 2019. PMID: 31143190 Free PMC article.
-
Autophagy is Involved in Neuroprotective Effect of Alpha7 Nicotinic Acetylcholine Receptor on Ischemic Stroke.Front Pharmacol. 2021 Apr 29;12:676589. doi: 10.3389/fphar.2021.676589. eCollection 2021. Front Pharmacol. 2021. PMID: 33995108 Free PMC article.
-
Targeting the α7 nicotinic acetylcholine receptor-A novel road towards the future treatment of skin diseases.Exp Dermatol. 2020 Sep;29(9):924-931. doi: 10.1111/exd.14173. Epub 2020 Sep 9. Exp Dermatol. 2020. PMID: 32780438 Review.
Cited by
-
Astrocyte-Neuron Interactions in Substance Use Disorders.Adv Neurobiol. 2024;39:165-191. doi: 10.1007/978-3-031-64839-7_7. Adv Neurobiol. 2024. PMID: 39190075 Review.
-
The synergistic effects of citicoline and silymarin on liver injury and thyroid hormone disturbances in γ-irradiated rats.Mol Biol Rep. 2025 Jan 30;52(1):176. doi: 10.1007/s11033-025-10255-2. Mol Biol Rep. 2025. PMID: 39883250
-
Molecular Docking Analysis at the Human α7-nAChR and Proliferative and Evoked-Calcium Changes in SH-SY5Y Cells by Imidacloprid and Acetamiprid Insecticides.Neurotox Res. 2024 Feb 20;42(2):16. doi: 10.1007/s12640-024-00697-0. Neurotox Res. 2024. PMID: 38376791
-
Neuroprotective Activities of Curcumin in Parkinson's Disease: A Review of the Literature.Int J Mol Sci. 2021 Oct 18;22(20):11248. doi: 10.3390/ijms222011248. Int J Mol Sci. 2021. PMID: 34681908 Free PMC article. Review.
-
Opinion: exploring alternative pathways to neuroprotection-nicotine and carbon monoxide as antioxidative factors in neurodegeneration and delirium.Front Neurol. 2025 Apr 9;16:1556456. doi: 10.3389/fneur.2025.1556456. eCollection 2025. Front Neurol. 2025. PMID: 40271113 Free PMC article. No abstract available.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
