This is a preprint.
Neutralizing activity of Sputnik V vaccine sera against SARS-CoV-2 variants
- PMID: 33851150
- PMCID: PMC8043464
- DOI: 10.21203/rs.3.rs-400230/v1
Neutralizing activity of Sputnik V vaccine sera against SARS-CoV-2 variants
Update in
-
Neutralizing activity of Sputnik V vaccine sera against SARS-CoV-2 variants.Nat Commun. 2021 Jul 26;12(1):4598. doi: 10.1038/s41467-021-24909-9. Nat Commun. 2021. PMID: 34312390 Free PMC article.
Abstract
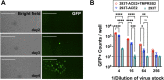
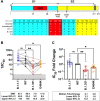
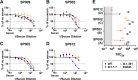
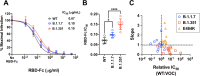
The novel pandemic betacoronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has infected at least 120 million people since its identification as the cause of a December 2019 viral pneumonia outbreak in Wuhan, China1,2. Despite the unprecedented pace of vaccine development, with six vaccines already in use worldwide, the emergence of SARS-CoV-2 'variants of concern' (VOC) across diverse geographic locales have prompted re-evaluation of strategies to achieve universal vaccination3. All three officially designated VOC carry Spike (S) polymorphisms thought to enable escape from neutralizing antibodies elicited during initial waves of the pandemic4-8. Here, we characterize the biological consequences of the ensemble of S mutations present in VOC lineages B.1.1.7 (501Y.V1) and B.1.351 (501Y.V2). Using a replication-competent EGFP-reporter vesicular stomatitis virus (VSV) system, rcVSV-CoV2-S, which encodes S from SARS coronavirus 2 in place of VSV-G, and coupled with a clonal HEK-293T ACE2 TMPRSS2 cell line optimized for highly efficient S-mediated infection, we determined that only 1 out of 12 serum samples from a cohort of recipients of the Gamaleya Sputnik V Ad26 / Ad5 vaccine showed effective neutralization (IC90) of rcVSV-CoV2-S: B.1.351 at full serum strength. The same set of sera efficiently neutralized S from B.1.1.7 and showed only moderately reduced activity against S carrying the E484K substitution alone. Taken together, our data suggest that control of some emergent SARS-CoV-2 variants may benefit from updated vaccines.
Conflict of interest statement
Figures
References
-
- COVID-19 Map - Johns Hopkins Coronavirus Resource Center. https://coronavirus.jhu.edu/map.html.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

