Assessment of Cumulative Incidence and Severity of Primary Open-Angle Glaucoma Among Participants in the Ocular Hypertension Treatment Study After 20 Years of Follow-up
- PMID: 33856434
- PMCID: PMC8050785
- DOI: 10.1001/jamaophthalmol.2021.0341
Assessment of Cumulative Incidence and Severity of Primary Open-Angle Glaucoma Among Participants in the Ocular Hypertension Treatment Study After 20 Years of Follow-up
Erratum in
-
Labeling Errors in Figure.JAMA Ophthalmol. 2021 Sep 1;139(9):1042. doi: 10.1001/jamaophthalmol.2021.2850. JAMA Ophthalmol. 2021. PMID: 34292316 Free PMC article. No abstract available.
Abstract
Importance: Ocular hypertension is an important risk factor for the development of primary open-angle glaucoma (POAG). Data from long-term follow-up can be used to inform the management of patients with ocular hypertension.
Objective: To determine the cumulative incidence and severity of POAG after 20 years of follow-up among participants in the Ocular Hypertension Treatment Study.
Design, setting, and participants: Participants in the Ocular Hypertension Treatment Study were followed up from February 1994 to December 2008 in 22 clinics. Data were collected after 20 years of follow-up (from January 2016 to April 2019) or within 2 years of death. Analyses were performed from July 2019 to December 2020.
Interventions: From February 28, 1994, to June 2, 2002 (phase 1), participants were randomized to receive either topical ocular hypotensive medication (medication group) or close observation (observation group). From June 3, 2002, to December 30, 2008 (phase 2), both randomization groups received medication. Beginning in 2009, treatment was no longer determined by study protocol. From January 7, 2016, to April 15, 2019 (phase 3), participants received ophthalmic examinations and visual function assessments.
Main outcomes and measures: Twenty-year cumulative incidence and severity of POAG in 1 or both eyes after adjustment for exposure time.
Results: A total of 1636 individuals (mean [SD] age, 55.4 [9.6] years; 931 women [56.9%]; 1138 White participants [69.6%]; 407 Black/African American participants [24.9%]) were randomized in phase 1 of the clinical trial. Of those, 483 participants (29.5%) developed POAG in 1 or both eyes (unadjusted incidence). After adjusting for exposure time, the 20-year cumulative incidence of POAG in 1 or both eyes was 45.6% (95% CI, 42.3%-48.8%) among all participants, 49.3% (95% CI, 44.5%-53.8%) among participants in the observation group, and 41.9% (95% CI, 37.2%-46.3%) among participants in the medication group. The 20-year cumulative incidence of POAG was 55.2% (95% CI, 47.9%-61.5%) among Black/African American participants and 42.7% (95% CI, 38.9%-46.3%) among participants of other races. The 20-year cumulative incidence for visual field loss was 25.2% (95% CI, 22.5%-27.8%). Using a 5-factor baseline model, the cumulative incidence of POAG among participants in the low-, medium-, and high-risk tertiles was 31.7% (95% CI, 26.4%-36.6%), 47.6% (95% CI, 41.6%-53.0%), and 59.8% (95% CI, 53.1%-65.5%), respectively.
Conclusions and relevance: In this study, only one-fourth of participants in the Ocular Hypertension Treatment Study developed visual field loss in either eye over long-term follow-up. This information, together with a prediction model, may help clinicians and patients make informed personalized decisions about the management of ocular hypertension.
Trial registration: ClinicalTrials.gov Identifier: NCT00000125.
Conflict of interest statement
Figures

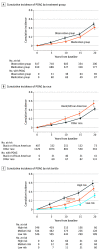
Comment in
-
Clinical Application of the 20-Year Results from the Ocular Hypertension Treatment Study (OHTS)-Reply.JAMA Ophthalmol. 2021 Oct 1;139(10):1146-1147. doi: 10.1001/jamaophthalmol.2021.3041. JAMA Ophthalmol. 2021. PMID: 34383004 No abstract available.
-
Clinical Application of the 20-Year Results from the Ocular Hypertension Treatment Study.JAMA Ophthalmol. 2021 Oct 1;139(10):1146. doi: 10.1001/jamaophthalmol.2021.3038. JAMA Ophthalmol. 2021. PMID: 34383028 No abstract available.
References
-
- Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701-713. doi: 10.1001/archopht.120.6.701 - DOI - PubMed
-
- Gordon MO, Torri V, Miglior S, et al. ; Ocular Hypertension Treatment Study Group; European Glaucoma Prevention Study Group . Validated prediction model for the development of primary open-angle glaucoma in individuals with ocular hypertension. Ophthalmology. 2007;114(1):10-19. doi: 10.1016/j.ophtha.2006.08.031 - DOI - PMC - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

