Ceramide kinase regulates TNF-α-induced immune responses in human monocytic cells
- PMID: 33859296
- PMCID: PMC8050074
- DOI: 10.1038/s41598-021-87795-7
Ceramide kinase regulates TNF-α-induced immune responses in human monocytic cells
Abstract
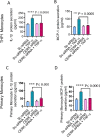
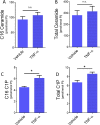
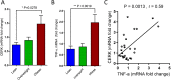
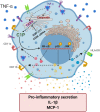
Ceramide kinase (CERK) phosphorylates ceramide to produce ceramide-1-phosphate (C1P), which is involved in the development of metabolic inflammation. TNF-α modulates inflammatory responses in monocytes associated with various inflammatory disorders; however, the underlying mechanisms remain not fully understood. Here, we investigated the role of CERK in TNF-α-induced inflammatory responses in monocytes. Our results show that disruption of CERK activity in monocytes, either by chemical inhibitor NVP-231 or by small interfering RNA (siRNA), results in the defective expression of inflammatory markers including CD11c, CD11b and HLA-DR in response to TNF-α. Our data show that TNF-α upregulates ceramide phosphorylation. Inhibition of CERK in monocytes significantly reduced the secretion of IL-1β and MCP-1. Similar results were observed in CERK-downregulated cells. TNF-α-induced phosphorylation of JNK, p38 and NF-κB was reduced by inhibition of CERK. Additionally, NF-κB/AP-1 activity was suppressed by the inhibition of CERK. Clinically, obese individuals had higher levels of CERK expression in PBMCs compared to lean individuals, which correlated with their TNF-α levels. Taken together, these results suggest that CERK plays a key role in regulating inflammatory responses in human monocytes during TNF-α stimulation. CERK may be a relevant target for developing novel therapies for chronic inflammatory diseases.
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Neutral sphingomyelinase 2 regulates inflammatory responses in monocytes/macrophages induced by TNF-α.Sci Rep. 2020 Oct 8;10(1):16802. doi: 10.1038/s41598-020-73912-5. Sci Rep. 2020. PMID: 33033337 Free PMC article.
-
Regulation and traffic of ceramide 1-phosphate produced by ceramide kinase: comparative analysis to glucosylceramide and sphingomyelin.J Biol Chem. 2008 Mar 28;283(13):8517-26. doi: 10.1074/jbc.M707107200. Epub 2007 Dec 16. J Biol Chem. 2008. PMID: 18086664
-
TNF-α Induces a Pro-Inflammatory Phenotypic Shift in Monocytes through ACSL1: Relevance to Metabolic Inflammation.Cell Physiol Biochem. 2019;52(3):397-407. doi: 10.33594/000000028. Epub 2019 Mar 8. Cell Physiol Biochem. 2019. PMID: 30845379
-
Novel signaling aspects of ceramide 1-phosphate.Biochim Biophys Acta Mol Cell Biol Lipids. 2020 Apr;1865(4):158630. doi: 10.1016/j.bbalip.2020.158630. Epub 2020 Jan 17. Biochim Biophys Acta Mol Cell Biol Lipids. 2020. PMID: 31958571 Review.
-
Ceramide kinase: the first decade.Cell Signal. 2011 Jun;23(6):999-1008. doi: 10.1016/j.cellsig.2010.11.012. Epub 2010 Nov 25. Cell Signal. 2011. PMID: 21111813 Review.
Cited by
-
Role of Ceramides in the Molecular Pathogenesis and Potential Therapeutic Strategies of Cardiometabolic Diseases: What we Know so Far.Front Cell Dev Biol. 2022 Jan 19;9:816301. doi: 10.3389/fcell.2021.816301. eCollection 2021. Front Cell Dev Biol. 2022. PMID: 35127726 Free PMC article. Review.
-
Lipidomics Analysis Deepen Understanding the Molecular Mechanisms in a Gouty Model Induced by Combination of MSU Crystals Injection and High-Fat Diet Feeding and the Intervention Mechanisms of Allopurinol.J Inflamm Res. 2024 Feb 13;17:1003-1018. doi: 10.2147/JIR.S443358. eCollection 2024. J Inflamm Res. 2024. PMID: 38370465 Free PMC article.
-
Fasting plasma methylglyoxal concentrations are associated with higher numbers of circulating intermediate and non-classical monocytes but with lower activation of intermediate monocytes: the Maastricht Study.J Endocrinol Invest. 2025 May;48(5):1257-1268. doi: 10.1007/s40618-025-02536-1. Epub 2025 Jan 23. J Endocrinol Invest. 2025. PMID: 39847265 Free PMC article.
-
The critical roles of bioactive sphingolipids in inflammation.J Biol Chem. 2025 Jul 11;301(8):110475. doi: 10.1016/j.jbc.2025.110475. Online ahead of print. J Biol Chem. 2025. PMID: 40653199 Free PMC article. Review.
-
Supplementation of Enriched Polyunsaturated Fatty Acids and CLA Cheese on High Fat Diet: Effects on Lipid Metabolism and Fat Profile.Foods. 2022 Jan 29;11(3):398. doi: 10.3390/foods11030398. Foods. 2022. PMID: 35159548 Free PMC article.
References
-
- Bajjalieh, S. M., Martin, T. F. & Floor, E. Synaptic vesicle ceramide kinase. A calcium-stimulated lipid kinase that co-purifies with brain synaptic vesicles. J. Biol. Chem.264, 14354–14360 (1989). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

