Lipophilicity Determines Routes of Uptake and Clearance, and Toxicity of an Alpha-Particle-Emitting Peptide Receptor Radiotherapy
- PMID: 33860213
- PMCID: PMC8033749
- DOI: 10.1021/acsptsci.1c00035
Lipophilicity Determines Routes of Uptake and Clearance, and Toxicity of an Alpha-Particle-Emitting Peptide Receptor Radiotherapy
Abstract
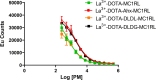
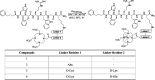
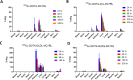
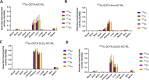
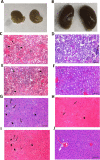
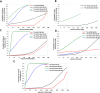
Lipophilicity is explored in the biodistribution (BD), pharmacokinetics (PK), radiation dosimetry (RD), and toxicity of an internally administered targeted alpha-particle therapy (TAT) under development for the treatment of metastatic melanoma. The TAT conjugate is comprised of the chelator DOTA (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate), conjugated to melanocortin receptor 1 specific peptidic ligand (MC1RL) using a linker moiety and chelation of the 225Ac radiometal. A set of conjugates were prepared with a range of lipophilicities (log D 7.4 values) by varying the chemical properties of the linker. Reported are the observations that higher log D 7.4 values are associated with decreased kidney uptake, decreased absorbed radiation dose, and decreased kidney toxicity of the TAT, and the inverse is observed for lower log D 7.4 values. Animals administered TATs with lower lipophilicities exhibited acute nephropathy and death, whereas animals administered the highest activity TATs with higher lipophilicities lived for the duration of the 7 month study and exhibited chronic progressive nephropathy. Changes in TAT lipophilicity were not associated with changes in liver uptake, dose, or toxicity. Significant observations include that lipophilicity correlates with kidney BD, the kidney-to-liver BD ratio, and weight loss and that blood urea nitrogen (BUN) levels correlated with kidney uptake. Furthermore, BUN was identified as having higher sensitivity and specificity of detection of kidney pathology, and the liver enzyme alkaline phosphatase (ALKP) had high sensitivity and specificity for detection of liver damage associated with the TAT. These findings suggest that tuning radiopharmaceutical lipophilicity can effectively modulate the level of kidney uptake to reduce morbidity and improve both safety and efficacy.
© 2021 American Chemical Society.
Conflict of interest statement
The authors declare the following competing financial interest(s): D.L.M. and N.K.T. are co-inventors of an awarded patent. D.L.M., T.J.W., M.L.M., H.K., and N.K.T. are co-inventors on a pending patent application. Modulation Therapeutics, Inc., has licensed related intellectual property, and M.L.M. is a co-founder of that company.
Figures

Similar articles
-
Biodistribution and Multicompartment Pharmacokinetic Analysis of a Targeted α Particle Therapy.Mol Pharm. 2020 Nov 2;17(11):4180-4188. doi: 10.1021/acs.molpharmaceut.0c00640. Epub 2020 Oct 1. Mol Pharm. 2020. PMID: 32960613
-
Melanocortin 1 Receptor-Targeted α-Particle Therapy for Metastatic Uveal Melanoma.J Nucl Med. 2019 Aug;60(8):1124-1133. doi: 10.2967/jnumed.118.217240. Epub 2019 Feb 7. J Nucl Med. 2019. PMID: 30733316 Free PMC article.
-
Development of Targeted Alpha Particle Therapy for Solid Tumors.Molecules. 2019 Nov 26;24(23):4314. doi: 10.3390/molecules24234314. Molecules. 2019. PMID: 31779154 Free PMC article. Review.
-
A gallium-labeled DOTA-alpha-melanocyte- stimulating hormone analog for PET imaging of melanoma metastases.J Nucl Med. 2004 Jan;45(1):116-23. J Nucl Med. 2004. PMID: 14734683
-
Targeted alpha therapy: a critical review of translational dosimetry research with emphasis on actinium-225.Q J Nucl Med Mol Imaging. 2020 Sep;64(3):265-277. doi: 10.23736/S1824-4785.20.03266-5. Epub 2020 May 21. Q J Nucl Med Mol Imaging. 2020. PMID: 32441067 Review.
Cited by
-
Structure-Activity Relationships and Biological Insights into PSMA-617 and Its Derivatives with Modified Lipophilic Linker Regions.ACS Omega. 2025 Feb 12;10(7):7077-7090. doi: 10.1021/acsomega.4c10142. eCollection 2025 Feb 25. ACS Omega. 2025. PMID: 40028088 Free PMC article.
-
eTFC-01: a dual-labeled chelate-bridged tracer for SSTR2-positive tumors.EJNMMI Radiopharm Chem. 2024 May 22;9(1):44. doi: 10.1186/s41181-024-00272-0. EJNMMI Radiopharm Chem. 2024. PMID: 38775990 Free PMC article.
-
Microbial Metabolites of 3-n-butylphthalide as Monoamine Oxidase A Inhibitors.Int J Mol Sci. 2023 Jun 25;24(13):10605. doi: 10.3390/ijms241310605. Int J Mol Sci. 2023. PMID: 37445788 Free PMC article.
-
64Cu2+ Complexes of Tripodal Amine Ligands' In Vivo Tumor and Liver Uptakes and Intracellular Cu Distribution in the Extrahepatic Bile Duct Carcinoma Cell Line TFK-1: A Basic Comparative Study.Pharmaceuticals (Basel). 2024 Jun 21;17(7):820. doi: 10.3390/ph17070820. Pharmaceuticals (Basel). 2024. PMID: 39065671 Free PMC article.
-
Evaluation of the Gly-Phe-Lys Linker to Reduce the Renal Radioactivity of a [64Cu]Cu-Labeled Multimeric cRGD Peptide.ACS Omega. 2025 Jan 24;10(4):4102-4120. doi: 10.1021/acsomega.4c10621. eCollection 2025 Feb 4. ACS Omega. 2025. PMID: 39926504 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
