Combined recombinase polymerase amplification/rkDNA-graphene oxide probing system for detection of SARS-CoV-2
- PMID: 33863409
- PMCID: PMC7973912
- DOI: 10.1016/j.aca.2021.338390
Combined recombinase polymerase amplification/rkDNA-graphene oxide probing system for detection of SARS-CoV-2
Abstract
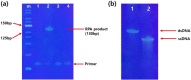
The development of rapid, highly sensitive, and selective methods for the diagnosis of infection by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) should help to prevent the spread of this pandemic virus. In this study, we combined recombinase polymerase amplification (RPA), as a means of isothermal DNA amplification, with an rkDNA-graphene oxide (GO) probe system to allow the rapid detection of SARS-CoV-2 with high sensitivity and selectivity. We used in situ enzymatic synthesis to prepare an rkDNA probe that was complementary to an RPA-amplified sequence of the target N-gene of SARS-CoV-2. The fluorescence of this rkDNA was perfectly quenched in the presence of GO. When the quenched rkDNA-GO system was added to the RPA-amplified sequence of the target SARS-CoV-2, the fluorescence recovered dramatically. The combined RPA/rkDNA-GO system exhibited extremely high selectivity (discrimination factor: 17.2) and sensitivity (LOD = 6.0 aM) for the detection of SARS-CoV-2. The total processing time was only 1.6 h. This combined RPA/rkDNA-GO system appears to be a very efficient and simple method for the point-of-care detection of SARS-CoV-2.
Keywords: Enzymatic in situ synthetic probe (rkDNA); Fluorescence; Graphene oxide; Isothermal amplification; RPA; SARS-CoV-2.
Copyright © 2021 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




Similar articles
-
Recent advances in recombinase polymerase amplification: Principle, advantages, disadvantages and applications.Front Cell Infect Microbiol. 2022 Nov 28;12:1019071. doi: 10.3389/fcimb.2022.1019071. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36519130 Free PMC article. Review.
-
rkDNA-graphene oxide as a simple probe for the rapid detection of miRNA21.Bioorg Med Chem Lett. 2020 Sep 1;30(17):127398. doi: 10.1016/j.bmcl.2020.127398. Epub 2020 Jul 15. Bioorg Med Chem Lett. 2020. PMID: 32738995
-
Isothermal recombinase polymerase amplification-lateral flow detection of SARS-CoV-2, the etiological agent of COVID-19.J Virol Methods. 2021 Oct;296:114227. doi: 10.1016/j.jviromet.2021.114227. Epub 2021 Jul 2. J Virol Methods. 2021. PMID: 34224752 Free PMC article.
-
Multiple ligation-Assisted recombinase polymerase amplification for highly sensitive and selective colorimetric detection of SARS-CoV-2.Talanta. 2023 Jan 15;252:123835. doi: 10.1016/j.talanta.2022.123835. Epub 2022 Aug 14. Talanta. 2023. PMID: 35985194 Free PMC article.
-
Application of recombinase polymerase amplification with lateral flow assay to pathogen point-of-care diagnosis.Front Cell Infect Microbiol. 2024 Nov 18;14:1475922. doi: 10.3389/fcimb.2024.1475922. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39624267 Free PMC article. Review.
Cited by
-
Rapid determination of SARS-CoV-2 nucleocapsid proteins based on 2D/2D MXene/P-BiOCl/Ru(bpy)32+ heterojunction composites to enhance electrochemiluminescence performance.Anal Chim Acta. 2022 Nov 22;1234:340522. doi: 10.1016/j.aca.2022.340522. Epub 2022 Oct 17. Anal Chim Acta. 2022. PMID: 36328721 Free PMC article.
-
Recent advances in recombinase polymerase amplification: Principle, advantages, disadvantages and applications.Front Cell Infect Microbiol. 2022 Nov 28;12:1019071. doi: 10.3389/fcimb.2022.1019071. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36519130 Free PMC article. Review.
-
Isothermal amplification-assisted diagnostics for COVID-19.Biosens Bioelectron. 2022 Jun 1;205:114101. doi: 10.1016/j.bios.2022.114101. Epub 2022 Feb 17. Biosens Bioelectron. 2022. PMID: 35202984 Free PMC article. Review.
-
Combining recombinase polymerase amplification and DNA-templated reaction for SARS-CoV-2 sensing with dual fluorescence and lateral flow assay output.Biopolymers. 2022 Apr;113(4):e23485. doi: 10.1002/bip.23485. Epub 2022 Jan 13. Biopolymers. 2022. PMID: 35023571 Free PMC article.
-
Dual-site ligation-assisted loop-mediated isothermal amplification (dLig-LAMP) for colorimetric and point-of-care determination of real SARS-CoV-2.Mikrochim Acta. 2022 Apr 5;189(5):176. doi: 10.1007/s00604-022-05293-7. Mikrochim Acta. 2022. PMID: 35381892 Free PMC article.
References
-
- Zhu Na, Zhang Dingyu, Wang Wenling, Li Xingwang, Yang Bo, Song Jingdong, Xiao Xiang, Huang Baoying, Shi Weifeng, Niu Roujian, Niu Peihua, Zhan Faxian, Ma Xuejun, Wang Dayan, Xu Wenbo, Wu Guizhen, Gao George F., Tan Wenjie. A novel coronavirus from patients with pneumonia in China. N. Engl. J. Med. 2019;382(2020):727–733. - PMC - PubMed
-
- Zhou Peng, Yang Xing-Lou, Wang Xian-Guang, Hu Ben, Zhang Lei, Zhang Wei, Hao-Rui Si, Zhu Yan, Li Bei, Huang Chao-Lin, Chen Hui-Dong, Chen Jing, Luo Yun, Guo Hua, Jiang Ren-Di, Liu Mei-Qin, Chen Ying, Shen Xu-Rui, Wang Xi, Zheng Xiao-Shuang, Zhao Kai, Chen Quan-Jiao, Deng Fei, Liu Lin-Lin, Yan Bing, Zhan Fa-Xian, Wang Yan-Yi, Xiao Geng-Fu, Shi Zheng-Li. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. - PMC - PubMed
-
- Corman Victor M., Rasche Andrea, Baronti Cecile, Aldabbagh Souhaib, Cadar Daniel, Chantal B., Reusken E.M., Pas Suzan D., Abraham Goorhuis, Schinkel Janke, Molenkamp Richard, Kümmerer Beate M., Tobias Bleicker, Brünink Sebastian, Eschbach-Bludau Monika, Anna M., Eis-Hübinger, Koopmans Marion P., Schmidt-Chanasit Jonas, Grobusch Martin P., de Lamballerie Xavier, Drosten Christian, Drexler Jan Felix. Assay optimization for molecular detection of Zika virus. Bull. World Health Organ. 2016;94(12):880–892. - PMC - PubMed
-
- Cao Lei, Cui Xingye, Hu Jie, Li Zedong, Choi Jane Ru, Yang Qingzhen, Lin Min, Li Ying Hui, Xu Feng. Advances in digital polymerase chain reaction (dPCR) and its emerging biomedical applications. Biosens. Bioelectron. 2017;90:459–474. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

