Trimetazidine Use in Parkinson's Disease: Is It a Resolved Problem?
- PMID: 33863783
- PMCID: PMC8143021
- DOI: 10.1523/ENEURO.0452-20.2021
Trimetazidine Use in Parkinson's Disease: Is It a Resolved Problem?
Abstract
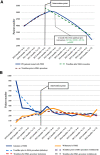
Trimetazidine (TMZ), an antianginal drug, can worsen the symptoms of movement disorders, therefore, the European Medicines Agency (EMA) recommended avoiding the use of this drug in Parkinson's disease (PD). We investigated the impact of this recommendation on the observed trend of TMZ use in PD in Hungary from 2010 to 2016 by conducting a nationwide, retrospective study of health administrative data of human subjects. Interrupted time series analyses were performed to explore changes in user trends after the EMA recommendations. We found that TMZ use in PD decreased by 6.56% in each six-month interval after the EMA intervention [a change in trend of -530.22, 95% confidence interval (CI) = -645.00 to -415.44, p < 0.001 and a decrease in level of -567.26, 95% CI = -910.99 to -223.53, p = 0.005 12 months postintervention]. TMZ discontinuation was the highest immediately after the intervention, however, its rate slowed down subsequently (a change in trend of -49.69, 95% CI = -85.14 to -14.24, p = 0.11 without significant level effects). The rate of new TMZ prescriptions did not reduce significantly, therefore, the decreased overall use was mainly attributable to the increased rate of discontinuation only. The main indications for TMZ use were circulatory system disorders, especially angina pectoris, however, off-label utilization was also considerable (40%). The EMA recommendations on TMZ use seem to be only moderately effective in Hungary. Although the number of patients with PD on the drug modestly decreased after the EMA restrictions, TMZ is still widely used in PD for both on-label and off-label indications.
Keywords: European Medicines Agency; Parkinson’s disease; angina pectoris; interrupted time series analysis; trimetazidine.
Copyright © 2021 Pintér et al.
Figures

References
-
- Cochrane Effective Practice and Organisation of Care (2017) Interrupted time series (ITS) analyses. EPOC Resources for Review Authors. Accessed December 16, 2019. Available from https://epoc.cochrane.org/sites/epoc.cochrane.org/files/public/uploads/R...
-
- Commission nationale de pharmacovigilance (2009) Compte rendu de la réunion du mardi 19 mai 2009. Accessed December 21, 2019. Available from: http://dev4-afssaps-marche2017.integra.fr/var/ansm_site/storage/original....
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
