Efficacy and safety of ravidasvir plus sofosbuvir in patients with chronic hepatitis C infection without cirrhosis or with compensated cirrhosis (STORM-C-1): interim analysis of a two-stage, open-label, multicentre, single arm, phase 2/3 trial
- PMID: 33865507
- PMCID: PMC9767645
- DOI: 10.1016/S2468-1253(21)00031-5
Efficacy and safety of ravidasvir plus sofosbuvir in patients with chronic hepatitis C infection without cirrhosis or with compensated cirrhosis (STORM-C-1): interim analysis of a two-stage, open-label, multicentre, single arm, phase 2/3 trial
Erratum in
-
Correction to Lancet Gastroenterol Hepatol 2021; 6: 448-58.Lancet Gastroenterol Hepatol. 2022 Aug;7(8):704. doi: 10.1016/S2468-1253(22)00209-6. Lancet Gastroenterol Hepatol. 2022. PMID: 35809604 Free PMC article. No abstract available.
Abstract
Background: In low-income and middle-income countries, affordable direct-acting antivirals are urgently needed to treat hepatitis C virus (HCV) infection. The combination of ravidasvir, a pangenotypic non-structural protein 5A (NS5A) inhibitor, and sofosbuvir has shown efficacy and safety in patients with chronic HCV genotype 4 infection. STORM-C-1 trial aimed to assess the efficacy and safety of ravidasvir plus sofosbuvir in a diverse population of adults chronically infected with HCV.
Methods: STORM-C-1 is a two-stage, open-label, phase 2/3 single-arm clinical trial in six public academic and non-academic centres in Malaysia and four public academic and non-academic centres in Thailand. Patients with HCV with compensated cirrhosis (Metavir F4 and Child-Turcotte-Pugh class A) or without cirrhosis (Metavir F0-3) aged 18-69 years were eligible to participate, regardless of HCV genotype, HIV infection status, previous interferon-based HCV treatment, or source of HCV infection. Once daily ravidasvir (200 mg) and sofosbuvir (400 mg) were prescribed for 12 weeks for patients without cirrhosis and for 24 weeks for those with cirrhosis. The primary endpoint was sustained virological response at 12 weeks after treatment (SVR12; defined as HCV RNA <12 IU/mL in Thailand and HCV RNA <15 IU/mL in Malaysia at 12 weeks after the end of treatment). This trial is registered with ClinicalTrials.gov, number NCT02961426, and the National Medical Research Register of Malaysia, NMRR-16-747-29183.
Findings: Between Sept 14, 2016, and June 5, 2017, 301 patients were enrolled in stage one of STORM-C-1. 98 (33%) patients had genotype 1a infection, 27 (9%) had genotype 1b infection, two (1%) had genotype 2 infection, 158 (52%) had genotype 3 infection, and 16 (5%) had genotype 6 infection. 81 (27%) patients had compensated cirrhosis, 90 (30%) had HIV co-infection, and 99 (33%) had received previous interferon-based treatment. The most common treatment-emergent adverse events were pyrexia (35 [12%]), cough (26 [9%]), upper respiratory tract infection (23 [8%]), and headache (20 [7%]). There were no deaths or treatment discontinuations due to serious adverse events related to study drugs. Of the 300 patients included in the full analysis set, 291 (97%; 95% CI 94-99) had SVR12. Of note, SVR12 was reported in 78 (96%) of 81 patients with cirrhosis and 153 (97%) of 158 patients with genotype 3 infection, including 51 (96%) of 53 patients with cirrhosis. There was no difference in SVR12 rates by HIV co-infection or previous interferon treatment.
Interpretation: In this first stage, ravidasvir plus sofosbuvir was effective and well tolerated in this diverse adult population of patients with chronic HCV infection. Ravidasvir plus sofosbuvir has the potential to provide an additional affordable, simple, and efficacious public health tool for large-scale implementation to eliminate HCV as a cause of morbidity and mortality.
Funding: National Science and Technology Development Agency, Thailand; Department of Disease Control, Ministry of Public Health, Thailand; Ministry of Health, Malaysia; UK Aid; Médecins Sans Frontières (MSF); MSF Transformational Investment Capacity; FIND; Pharmaniaga; Starr International Foundation; Foundation for Art, Research, Partnership and Education; and the Swiss Agency for Development and Cooperation.
Copyright © 2021 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Figures
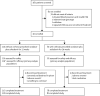

Comment in
-
Affordable treatment and political commitment are crucial to eliminate hepatitis C globally.Lancet Gastroenterol Hepatol. 2021 Jun;6(6):414-416. doi: 10.1016/S2468-1253(21)00135-7. Epub 2021 Apr 16. Lancet Gastroenterol Hepatol. 2021. PMID: 33865506 Free PMC article. No abstract available.
References
-
- WHO HCV Factsheet. 2019. https://www.who.int/news-room/fact-sheets/detail/hepatitis-c
-
- WHO Global health sector strategy on viral hepatitis 2016-2021. 2016. https://www.who.int/hepatitis/strategy2016-2021/ghss-hep/en/
-
- Gamkrelidze I, Pawlotsky J, Lazarus J V, et al. Progress towards hepatitis C virus elimination in high-income countries: an updated analysis. Liver Int. 2021;41:456–463. - PubMed
-
- Noronha M. Endgame: Egypt's path to eliminating hepatitis B and C. 2019. https://eiuperspectives.economist.com/healthcare/e
-
- WHO Global report on access to hepatitis C treatment. Focus on overcoming barriers. 2016. https://www.who.int/hepatitis/.publications/hep-c-access-report/en/
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

