Liraglutide, a TFEB-Mediated Autophagy Agonist, Promotes the Viability of Random-Pattern Skin Flaps
- PMID: 33868571
- PMCID: PMC8032515
- DOI: 10.1155/2021/6610603
Liraglutide, a TFEB-Mediated Autophagy Agonist, Promotes the Viability of Random-Pattern Skin Flaps
Abstract
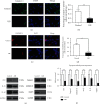
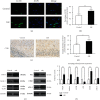
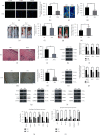
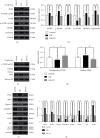
Random skin flaps are commonly used in reconstruction surgery. However, distal necrosis of the skin flap remains a difficult problem in plastic surgery. Many studies have shown that activation of autophagy is an important means of maintaining cell homeostasis and can improve the survival rate of flaps. In the current study, we investigated whether liraglutide can promote the survival of random flaps by stimulating autophagy. Our results show that liraglutide can significantly improve flap viability, increase blood flow, and reduce tissue oedema. In addition, we demonstrated that liraglutide can stimulate angiogenesis and reduce pyroptosis and oxidative stress. Through immunohistochemistry analysis and Western blotting, we verified that liraglutide can enhance autophagy, while the 3-methylladenine- (3MA-) mediated inhibition of autophagy enhancement can significantly reduce the benefits of liraglutide described above. Mechanistically, we showed that the ability of liraglutide to enhance autophagy is mediated by the activation of transcription factor EB (TFEB) and its subsequent entry into the nucleus to activate autophagy genes, a phenomenon that may result from AMPK-MCOLN1-calcineurin signalling pathway activation. Taken together, our results show that liraglutide is an effective drug that can significantly improve the survival rate of random flaps by enhancing autophagy, inhibiting oxidative stress in tissues, reducing pyroptosis, and promoting angiogenesis, which may be due to the activation of TFEB via the AMPK-MCOLN1-calcineurin signalling pathway.
Copyright © 2021 Xuwei Zhu et al.
Conflict of interest statement
The authors have declared that no competing interest exists.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

