Subinhibitory Concentrations of Biogenic Silver Nanoparticles Affect Motility and Biofilm Formation in Pseudomonas aeruginosa
- PMID: 33869087
- PMCID: PMC8047417
- DOI: 10.3389/fcimb.2021.656984
Subinhibitory Concentrations of Biogenic Silver Nanoparticles Affect Motility and Biofilm Formation in Pseudomonas aeruginosa
Abstract
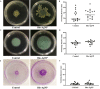
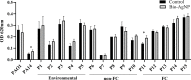
Biogenic silver nanoparticles (bio-AgNPs) are increasingly recognized as an antibiofilm and antivirulence strategy against P. aeruginosa, a bacterium that causes chronic infections in immunocompromised and cystic fibrosis patients. This study aimed to investigate the effects of subinhibitory concentrations of bio-AgNPs on motility and biofilm formation in P. aeruginosa. Bio-AgNPs were synthesized via reduction of ionic silver catalyzed by cell-free culture filtrate from Fusarium oxysporum. A total of 17 P. aeruginosa isolates and strains were evaluated for swarming, swimming, and twitching motility in the presence and absence (control) of bio-AgNPs, including 10 clinical isolates from patients with and without cystic fibrosis, 5 environmental isolates obtained from the public water supply system, and 2 reference strains (PAO1 and PA14). Isolates were identified by biochemical and molecular methods. Minimum inhibitory concentrations (MICs) were determined by the broth microdilution method. Swarming, swimming, and twitching motility assays were performed in Petri dishes. Biofilm formation capacity was assessed quantitatively by the crystal violet method. MIC values ranged from 15.62 to 62.50 µM. The results showed that subinhibitory concentrations of bio-AgNPs (½ MIC, 7.81-31.25 µM) significantly increased (p < 0.05) swarming, swimming, and twitching motility in 40.0, 40.0, and 46.7% of isolates, respectively. Subinhibitory bio-AgNP treatment enhanced (p < 0.05) biofilm formation capacity in PA14 and a cystic fibrosis isolate (P11). It is concluded that subinhibitory concentrations of bio-AgNPs increased biofilm formation and swarming, swimming, and twitching motility in PA14 and some P. aeruginosa isolates. These virulence factors are directly involved with quorum-sensing systems. Further research should investigate the effects of AgNPs on P. aeruginosa quorum sensing to help elucidate their mechanism of action at subinhibitory concentrations.
Keywords: antibiofilm; antivirulence; metal nanoparticle; quorum sensing; virulence factor.
Copyright © 2021 Saeki, Yamada, de Araujo, Anversa, Garcia, de Souza, Martins, Kobayashi and Nakazato.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Ahmed S. A. K. S., Rudden M., Smyth T. J., Dooley J. S. G., Marchant R., Banat I. M. (2019). Natural quorum sensing inhibitors effectively downregulate gene expression of Pseudomonas aeruginosa virulence factors. Appl. Microbiol. Biotechnol. 103 (8), 3521–3535. 10.1007/s00253-019-09618-0 - DOI - PMC - PubMed
-
- Ahmed T., Pattnaik S., Khan M. B., Ampasala D. R., Busi S., Sarma V. V. (2020). Inhibition of quorum sensing-associated virulence factors and biofilm formation in Pseudomonas aeruginosa PAO1 by Mycoleptodiscus indicus PUTY1. Braz. J. Microbiol. 51 (2), 467–487. 10.1007/s42770-020-00235-y - DOI - PMC - PubMed
-
- Anversa L., Stancari R. C. A., Garbelotti M., Ruiz L. S., Pereira V. B. R., Nascentes G. A. N., et al. (2019). Pseudomonas aeruginosa in public water supply. Water Pract. Technol. 14 (3), 732–737. 10.2166/wpt.2019.057 - DOI
-
- Bahramian A., Khoshnood S., Shariati A., Doustdar F., Chirani A. S., Heidary M. (2019). Molecular characterization of the pilS2 gene and its association with the frequency of Pseudomonas aeruginosa plasmid pKLC102 and PAPI-1 pathogenicity island. Infect. Drug Resist. 12, 221–227. 10.2147/IDR.S188527 - DOI - PMC - PubMed
-
- CLSI (2015). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard—Tenth Edition. CLSI document M07-A10. Wayne, PA: Clinical and Laboratory Standards Institute.
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

