Antibacterial Albumin-Tannic Acid Coatings for Scaffold-Guided Breast Reconstruction
- PMID: 33869154
- PMCID: PMC8044405
- DOI: 10.3389/fbioe.2021.638577
Antibacterial Albumin-Tannic Acid Coatings for Scaffold-Guided Breast Reconstruction
Abstract
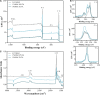
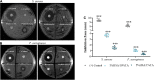
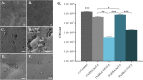
Infection is the major cause of morbidity after breast implant surgery. Biodegradable medical-grade polycaprolactone (mPCL) scaffolds designed and rooted in evidence-based research offer a promising alternative to overcome the limitations of routinely used silicone implants for breast reconstruction. Nevertheless, as with any implant, biodegradable scaffolds are susceptible to bacterial infection too, especially as bacteria can rapidly colonize the biomaterial surface and form biofilms. Biofilm-related infections are notoriously challenging to treat and can lead to chronic infection and persisting inflammation of surrounding tissue. To date, no clinical solution that allows to efficiently prevent bacterial infection while promoting correct implant integration, has been developed. In this study, we demonstrated for the first time, to our knowledge that the physical immobilization of 1 and 5% human serum albumin (HSA) onto the surface of 3D printed macro- and microporous mPCL scaffolds, resulted in a reduction of Staphylococcus aureus colonization by 71.7 ± 13.6% and 54.3 ± 12.8%, respectively. Notably, when treatment of scaffolds with HSA was followed by tannic acid (TA) crosslinking/stabilization, uniform and stable coatings with improved antibacterial activity were obtained. The HSA/TA-coated scaffolds were shown to be stable when incubated at physiological conditions in cell culture media for 7 days. Moreover, they were capable of inhibiting the growth of S. aureus and Pseudomonas aeruginosa, two most commonly found bacteria in breast implant infections. Most importantly, 1%HSA/10%TA- and 5%HSA/1%TA-coated scaffolds were able to reduce S. aureus colonization on the mPCL surface, by 99.8 ± 0.1% and 98.8 ± 0.6%, respectively, in comparison to the non-coated control specimens. This system offers a new biomaterial strategy to effectively translate the prevention of biofilm-related infections on implant surfaces without relying on the use of prophylactic antibiotic treatment.
Keywords: 3D printing; albumin; antibacterial coating; bacterial infection; polycaprolactone; scaffold; tannic acid.
Copyright © 2021 Cometta, Bock, Suresh, Dargaville and Hutmacher.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Unravelling the physicochemical and antimicrobial mechanisms of human serum albumin/tannic acid coatings for medical-grade polycaprolactone scaffolds.Bioact Mater. 2024 Aug 28;42:68-84. doi: 10.1016/j.bioactmat.2024.08.023. eCollection 2024 Dec. Bioact Mater. 2024. PMID: 39280579 Free PMC article.
-
Melimine-Modified 3D-Printed Polycaprolactone Scaffolds for the Prevention of Biofilm-Related Biomaterial Infections.ACS Nano. 2022 Oct 25;16(10):16497-16512. doi: 10.1021/acsnano.2c05812. Epub 2022 Oct 16. ACS Nano. 2022. PMID: 36245096 Free PMC article.
-
Novel Antibiotic-loaded Point-of-care Implant Coating Inhibits Biofilm.Clin Orthop Relat Res. 2015 Jul;473(7):2270-82. doi: 10.1007/s11999-014-4130-8. Clin Orthop Relat Res. 2015. PMID: 25604874 Free PMC article.
-
Biofilm in implant infections: its production and regulation.Int J Artif Organs. 2005 Nov;28(11):1062-8. doi: 10.1177/039139880502801103. Int J Artif Organs. 2005. PMID: 16353112 Review.
-
Surface treatment strategies to combat implant-related infection from the beginning.J Orthop Translat. 2018 Sep 28;17:42-54. doi: 10.1016/j.jot.2018.09.001. eCollection 2019 Apr. J Orthop Translat. 2018. PMID: 31194031 Free PMC article. Review.
Cited by
-
Bone Grafting with Albumin-Impregnated Bone Allograft After Odontogenic Cyst Removal.J Clin Med. 2025 Jun 12;14(12):4173. doi: 10.3390/jcm14124173. J Clin Med. 2025. PMID: 40565919 Free PMC article.
-
Assessing the Biocompatibility of Tannic Acid-Based Biomaterials: Addressing Challenges in Standard Cytotoxic Assays.Bioengineering (Basel). 2025 Jun 16;12(6):660. doi: 10.3390/bioengineering12060660. Bioengineering (Basel). 2025. PMID: 40564476 Free PMC article.
-
Albumin: A Review of Market Trends, Purification Methods, and Biomedical Innovations.Curr Issues Mol Biol. 2025 Apr 26;47(5):303. doi: 10.3390/cimb47050303. Curr Issues Mol Biol. 2025. PMID: 40699702 Free PMC article. Review.
-
Unravelling the physicochemical and antimicrobial mechanisms of human serum albumin/tannic acid coatings for medical-grade polycaprolactone scaffolds.Bioact Mater. 2024 Aug 28;42:68-84. doi: 10.1016/j.bioactmat.2024.08.023. eCollection 2024 Dec. Bioact Mater. 2024. PMID: 39280579 Free PMC article.
-
Films, Gels and Electrospun Fibers from Serum Albumin Globular Protein for Medical Device Coating, Biomolecule Delivery and Regenerative Engineering.Pharmaceutics. 2022 Oct 27;14(11):2306. doi: 10.3390/pharmaceutics14112306. Pharmaceutics. 2022. PMID: 36365125 Free PMC article. Review.
References
-
- Ackbarow T., Buehler M. (2007). Hierarchical coexistence of universality and diversity controls robustness and multi-functionality in intermediate filament protein networks. Nat. Preced. 2 1–1. 10.1038/npre.2007.826.1 - DOI
-
- Aguilera J. R., Venegas V., Oliva J. M., Sayagués M. J., de Miguel M., Sánchez-Alcázar J. A., et al. (2016). Targeted multifunctional tannic acid nanoparticles. RSC Adv. 6 7279–7287. 10.1039/C5RA19405A - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

