Innate Lymphoid Cells as Regulators of Epithelial Integrity: Therapeutic Implications for Inflammatory Bowel Diseases
- PMID: 33869257
- PMCID: PMC8044918
- DOI: 10.3389/fmed.2021.656745
Innate Lymphoid Cells as Regulators of Epithelial Integrity: Therapeutic Implications for Inflammatory Bowel Diseases
Abstract
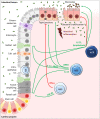
The occurrence of epithelial defects in the gut relevantly contributes to the pathogenesis of inflammatory bowel diseases (IBD), whereby the impairment of intestinal epithelial barrier integrity seems to represent a primary trigger as well as a disease amplifying consequence of the chronic inflammatory process. Besides epithelial cell intrinsic factors, accumulated and overwhelmingly activated immune cells and their secretome have been identified as critical modulators of the pathologically altered intestinal epithelial cell (IEC) function in IBD. In this context, over the last 10 years increasing levels of attention have been paid to the group of innate lymphoid cells (ILCs). This is in particular due to a preferential location of these rather newly described innate immune cells in close proximity to mucosal barriers, their profound capacity to secrete effector cytokines and their numerical and functional alteration under chronic inflammatory conditions. Aiming on a comprehensive and updated summary of our current understanding of the bidirectional mucosal crosstalk between ILCs and IECs, this review article will in particular focus on the potential capacity of gut infiltrating type-1, type-2, and type-3 helper ILCs (ILC1s, ILC2s, and ILC3s, respectively) to impact on the survival, differentiation, and barrier function of IECs. Based on data acquired in IBD patients or in experimental models of colitis, we will discuss whether the different ILC subgroups could serve as potential therapeutic targets for maintenance of epithelial integrity and/or mucosal healing in IBD.
Keywords: ILC plasticity; colitis; cytokines; inflammatory bowel diseases; innate lymphoid cells; intestinal epithelium.
Copyright © 2021 Schulz-Kuhnt, Neurath, Wirtz and Atreya.
Conflict of interest statement
MN has served as an advisor for Pentax, Giuliani, MSD, Abbvie, Janssen, Takeda and Boehringer. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Cross-talk between type 3 innate lymphoid cells and the gut microbiota in inflammatory bowel disease.Curr Opin Gastroenterol. 2015 Nov;31(6):449-55. doi: 10.1097/MOG.0000000000000217. Curr Opin Gastroenterol. 2015. PMID: 26398682 Free PMC article. Review.
-
Innate Lymphoid Cells in Intestinal Inflammation.Front Immunol. 2017 Oct 13;8:1296. doi: 10.3389/fimmu.2017.01296. eCollection 2017. Front Immunol. 2017. PMID: 29081776 Free PMC article. Review.
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
-
Innate Lymphoid Cells in Intestinal Homeostasis and Inflammatory Bowel Disease.Int J Mol Sci. 2021 Jul 16;22(14):7618. doi: 10.3390/ijms22147618. Int J Mol Sci. 2021. PMID: 34299236 Free PMC article. Review.
-
Helper-Like Type-1 Innate Lymphoid Cells in Inflammatory Bowel Disease.Front Immunol. 2022 Jun 23;13:903688. doi: 10.3389/fimmu.2022.903688. eCollection 2022. Front Immunol. 2022. PMID: 35844597 Free PMC article. Review.
Cited by
-
Probiotics for the treatment of ulcerative colitis: a review of experimental research from 2018 to 2022.Front Microbiol. 2023 Jul 6;14:1211271. doi: 10.3389/fmicb.2023.1211271. eCollection 2023. Front Microbiol. 2023. PMID: 37485519 Free PMC article. Review.
-
Probiotics in Intestinal Mucosal Healing: A New Therapy or an Old Friend?Pharmaceuticals (Basel). 2021 Nov 19;14(11):1181. doi: 10.3390/ph14111181. Pharmaceuticals (Basel). 2021. PMID: 34832962 Free PMC article. Review.
-
The Inflammatory Bowel Disease Transcriptome and Metatranscriptome Meta-Analysis (IBD TaMMA) framework.Nat Comput Sci. 2021 Aug;1(8):511-515. doi: 10.1038/s43588-021-00114-y. Epub 2021 Aug 20. Nat Comput Sci. 2021. PMID: 38217242 Free PMC article.
-
Innate immune cellular therapeutics in transplantation.Front Transplant. 2023;2:1067512. doi: 10.3389/frtra.2023.1067512. Epub 2023 Mar 31. Front Transplant. 2023. PMID: 37994308 Free PMC article.
-
Pathophysiology of Inflammatory Bowel Disease: Innate Immune System.Int J Mol Sci. 2023 Jan 12;24(2):1526. doi: 10.3390/ijms24021526. Int J Mol Sci. 2023. PMID: 36675038 Free PMC article. Review.
References
-
- Alatab S, Sepanlou SG, Ikuta K, Vahedi H, Bisignano C, Safiri S, et al. . The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. (2020) 5:17–30. 10.1016/S2468-1253(19)30333-4 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

