Real-World Effectiveness of Human Papillomavirus Vaccination Against Cervical Cancer
- PMID: 33876216
- PMCID: PMC8486335
- DOI: 10.1093/jnci/djab080
Real-World Effectiveness of Human Papillomavirus Vaccination Against Cervical Cancer
Abstract
Background: The primary goal of human papillomavirus (HPV) vaccination is to reduce morbidity and mortality from HPV-associated disease, especially cervical cancer. We determined the real-world effectiveness of HPV vaccination against cervical cancer.
Methods: The study included women aged 17-30 years living in Denmark October 2006-December 2019. From nationwide registries, information on HPV vaccination and cervical cancer diagnoses were retrieved. Incidence rate ratios (IRRs) with 95% confidence intervals (CIs) for cervical cancer according to vaccination status were estimated using Poisson regression with HPV vaccination treated as a time-varying variable and stratified by age at vaccination. We adjusted for attained age, education, and ethnicity. To address the effect of prevalent disease, different buffer periods were used, with 1-year buffer period as primary analysis.
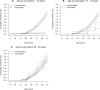
Results: The cohort comprised 867 689 women. At baseline, 36.3% were vaccinated at age 16 years and younger, and during follow-up, 19.3% and 2.3% were vaccinated at ages 17-19 years and 20-30 years, respectively. For women vaccinated at ages 16 years and younger or 17-19 years, the IRRs of cervical cancer were 0.14 (95% CI = 0.04 to 0.53) and 0.32 (95% CI = 0.08 to 1.28), respectively, compared with unvaccinated women. In women aged 20-30 years at vaccination, the incidence rate was higher than among unvaccinated women (IRR = 1.19, 95% CI = 0.80 to 1.79) but slightly decreased with increasing buffer period (IRR = 0.85, 95% CI = 0.55 to 1.32, with 4-year buffer period).
Conclusion: HPV vaccine effectiveness against cervical cancer at the population level is high among girls vaccinated younger than age 20 years. The lack of immediate effect in women vaccinated at age 20-30 years points to the importance of early age at vaccination.
© The Author(s) 2021. Published by Oxford University Press. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures

Comment in
-
Real-World HPV Vaccine Effectiveness Studies: Guideposts for Interpretation of Current and Future Studies.J Natl Cancer Inst. 2021 Oct 1;113(10):1270-1271. doi: 10.1093/jnci/djab081. J Natl Cancer Inst. 2021. PMID: 33876237 No abstract available.
References
-
- Serrano B, de Sanjosé S, Tous S, et al. Human papillomavirus genotype distribution for HPVs 6, 11, 16, 18, 31, 33, 45, 52 and 58 in female anogenital lesions. Eur J Cancer. 2015;51(13):1732–1741. - PubMed
-
- Walboomers JM, Jacobs MV, Manos MM, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189(1):12–19. - PubMed
-
- World Health Organization. A global strategy for elimination of cervical cancer; 2020. . https://www.who.int/publications/i/item/9789240014107. Accessed February 25, 2021.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

