Clinicians' knowledge and attitudes towards patient reported outcomes in colorectal cancer care - insights from qualitative interviews
- PMID: 33879141
- PMCID: PMC8056693
- DOI: 10.1186/s12913-021-06361-z
Clinicians' knowledge and attitudes towards patient reported outcomes in colorectal cancer care - insights from qualitative interviews
Abstract
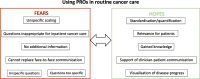
Introduction: Patient-reported outcomes (PROs) can be used in cancer care to monitor patients' disease-related symptoms and functional status. However, successful implementation of such instruments is only possible if clinical staff are convinced of the clinical benefits. It is therefore crucial to investigate the attitudes of clinical staff to PROs in routine cancer care.
Methods: Semi-structured, guideline-based interviews were held with 12 clinicians working in certified colorectal cancer centers in Germany who are taking part in an observational study on PROs (five surgeons, two oncologists, one psycho-oncologist, two oncological care nurses, one stoma therapist, and one physician assistant) in order to investigate firstly, how clinicians describe PRO instruments ("wording"); and secondly, the clinicians' general attitude toward PROs. A qualitative content analysis according to Kuckartz was performed.
Results: The wording used to describe PROs was not consistent. Statements on attitudes toward PROs were very heterogeneous and were therefore categorized into "(rather) positive" and "(rather) negative." The principal advantages of PROs mentioned by participants included broader, structured knowledge about patients and treatment, as well as relevance for patients. Subcategories for (rather) negative attitudes included statements expressing doubts about the questionnaires and "no need for PROs."
Discussion: The clinicians participating mainly expressed fairly positive attitudes toward PROs. However, they had little knowledge about PROs in general and the interviews therefore mainly reflect their expectations and assumptions about them. These initial impressions may be regarded as providing a basis for future implementation strategies and for training of clinicians on how to use PROs in routine cancer care.
Keywords: Cancer care; EORTC; Implementation; Patient-reported outcome measures; Patient-reported outcomes; Routine care.
Conflict of interest statement
All authors expect for HP are employees of the German Cancer Society.
Figures
Similar articles
-
Could existing infrastructure for using patient-reported outcomes as quality measures also be used for individual care in patients with colorectal cancer?BMC Health Serv Res. 2021 May 11;21(1):448. doi: 10.1186/s12913-021-06457-6. BMC Health Serv Res. 2021. PMID: 33975586 Free PMC article.
-
Implementation analysis of patient reported outcomes (PROs) in oncological routine care: an observational study protocol.Health Qual Life Outcomes. 2020 Jan 2;18(1):3. doi: 10.1186/s12955-019-1262-2. Health Qual Life Outcomes. 2020. PMID: 31898544 Free PMC article.
-
Clinicians' perspectives on the integration of electronic patient-reported outcomes into dermatology clinics: a qualitative study.Qual Life Res. 2022 Jun;31(6):1719-1725. doi: 10.1007/s11136-021-03030-w. Epub 2021 Nov 2. Qual Life Res. 2022. PMID: 34727299
-
Examining perspectives on the adoption and use of computer-based patient-reported outcomes among clinicians and health professionals: a Q methodology study.J Am Med Inform Assoc. 2022 Jan 29;29(3):443-452. doi: 10.1093/jamia/ocab257. J Am Med Inform Assoc. 2022. PMID: 34871423 Free PMC article. Review.
-
[Psychometric characteristics of questionnaires designed to assess the knowledge, perceptions and practices of health care professionals with regards to alcoholic patients].Encephale. 2004 Sep-Oct;30(5):437-46. doi: 10.1016/s0013-7006(04)95458-9. Encephale. 2004. PMID: 15627048 Review. French.
Cited by
-
Different Approaches for Case-Mix Adjustment of Patient-Reported Outcomes to Compare Healthcare Providers-Methodological Results of a Systematic Review.Cancers (Basel). 2021 Aug 5;13(16):3964. doi: 10.3390/cancers13163964. Cancers (Basel). 2021. PMID: 34439117 Free PMC article. Review.
-
Patient-Reported Outcomes in Colorectal Surgery.Clin Colon Rectal Surg. 2023 Feb 8;36(4):240-251. doi: 10.1055/s-0043-1761607. eCollection 2023 Jul. Clin Colon Rectal Surg. 2023. PMID: 37223227 Free PMC article. Review.
-
Exploring the use of health and wellbeing measures during pregnancy and the first year following birth in women living with pre-existing long-term conditions: qualitative interviews with women and healthcare professionals.BMC Health Serv Res. 2021 Jun 24;21(1):597. doi: 10.1186/s12913-021-06615-w. BMC Health Serv Res. 2021. PMID: 34162368 Free PMC article.
-
Functional Results After Nerve-Sparing, Sphincter Preserving Rectal Cancer Surgery: Patient-Reported Outcomes of Sexual and Urinary Dysfunction.Indian J Surg Oncol. 2023 Dec;14(4):868-875. doi: 10.1007/s13193-023-01794-w. Epub 2023 Jul 7. Indian J Surg Oncol. 2023. PMID: 38187835 Free PMC article.
-
Collection and Utilization of Patient-Reported Outcome Measures in a Colorectal Surgery Clinic.J Surg Res. 2022 Dec;280:515-525. doi: 10.1016/j.jss.2022.07.042. Epub 2022 Sep 5. J Surg Res. 2022. PMID: 36081311 Free PMC article.
References
-
- Denis F, Yossi S, Septans A-L, Charron A, Voog E, Dupuis O, Ganem G, Pointreau Y, Letellier C. Improving survival in patients treated for a lung cancer using self-evaluated symptoms reported through a web application. Am J Clin Oncol. 2017;40(5):464–469. doi: 10.1097/COC.0000000000000189. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous