SLC22A14 is a mitochondrial riboflavin transporter required for sperm oxidative phosphorylation and male fertility
- PMID: 33882315
- PMCID: PMC8065176
- DOI: 10.1016/j.celrep.2021.109025
SLC22A14 is a mitochondrial riboflavin transporter required for sperm oxidative phosphorylation and male fertility
Abstract
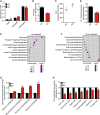
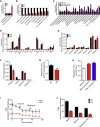
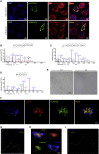
Ablation of Slc22a14 causes male infertility in mice, but the underlying mechanisms remain unknown. Here, we show that SLC22A14 is a riboflavin transporter localized at the inner mitochondrial membrane of the spermatozoa mid-piece and show by genetic, biochemical, multi-omic, and nutritional evidence that riboflavin transport deficiency suppresses the oxidative phosphorylation and reprograms spermatozoa energy metabolism by disrupting flavoenzyme functions. Specifically, we find that fatty acid β-oxidation (FAO) is defective with significantly reduced levels of acyl-carnitines and metabolites from the TCA cycle (the citric acid cycle) but accumulated triglycerides and free fatty acids in Slc22a14 knockout spermatozoa. We demonstrate that Slc22a14-mediated FAO is essential for spermatozoa energy generation and motility. Furthermore, sperm from wild-type mice treated with a riboflavin-deficient diet mimics those in Slc22a14 knockout mice, confirming that an altered riboflavin level causes spermatozoa morphological and bioenergetic defects. Beyond substantially advancing our understanding of spermatozoa energy metabolism, our study provides an attractive target for the development of male contraceptives.
Keywords: SLC22A14 transporter; energy metabolism; fatty acid β-oxidation; male infertility; riboflavin.
Copyright © 2021 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing conflicts of interest.
Figures





References
-
- Amaral A., Lourenço B., Marques M., Ramalho-Santos J. Mitochondria functionality and sperm quality. Reproduction. 2013;146:R163–R174. - PubMed
-
- Am-in N., Kirkwood R.N., Techakumphu M., Tantasuparuk W. Lipid profiles of sperm and seminal plasma from boars having normal or low sperm motility. Theriogenology. 2011;75:897–903. - PubMed
-
- Asghari A., Marashi S.A., Ansari-Pour N. A sperm-specific proteome-scale metabolic network model identifies non-glycolytic genes for energy deficiency in asthenozoospermia. Syst Biol Reprod Med. 2017;63:100–112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

