Actin-Like Protein 8 Promotes the Progression of Triple-Negative Breast Cancer via Activating PI3K/AKT/mTOR Pathway
- PMID: 33883901
- PMCID: PMC8053609
- DOI: 10.2147/OTT.S291403
Actin-Like Protein 8 Promotes the Progression of Triple-Negative Breast Cancer via Activating PI3K/AKT/mTOR Pathway
Abstract
Objective: The purpose of this study was to investigate the function of actin-like protein 8 (ACTL8) on triple-negative breast cancer (TNBC) and its potential mechanisms.
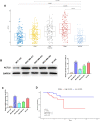
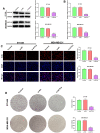
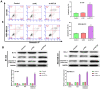
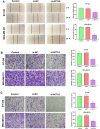
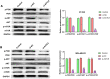
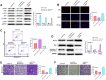
Methods: In our study, ACTL8 expression and the prognostic values of ACTL8 were evaluated via the dataset from the Cancer Genome Atlas (TCGA). At the same time, the expression of ACTL8 in TNBC cells was measured by Western blot and qRT-PCR. Then, the effects of ACTL8 on the growth and metastasis of TNBC were investigated by using 5-ethynyl-20-deoxyuridine (EdU), colony formation, flow cytometry, wound healing and transwell assays. Mechanistically, Western blot was performed to confirm the interaction between ACTL8 and phosphatidylinositol 3'-kinase/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR) signaling pathway in TNBC.
Results: ACTL8 expression was upregulated in TNBC and associated with the poor prognosis of TNBC. Silencing ACTL8 suppressed the proliferation, migration and invasion, also promoted the apoptosis in MDA-MB-231 and BT-549 cells. Moreover, we found that silencing ACTL8 could inhibit the activation of PI3K/AKT/mTOR signaling pathway in MDA-MB-231 and BT-549 cells. Meanwhile, the impact of silencing ACTL8 on the proliferation, apoptosis, migration and invasion was enhanced by PI3K/AKT/mTOR pathway inhibitor (Wortmannin) and reversed by PI3K/AKT/mTOR pathway activator (740Y-P).
Conclusion: Our data demonstrated that ACTL8 may facilitate the proliferation, migration and invasion, while inhibiting apoptosis through activating PI3K/Akt/mTOR signaling pathway in TNBC.
Keywords: ACTL8; PI3K/AKT/mTOR pathway; TNBC; invasion; migration; proliferation.
© 2021 Fan et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
Similar articles
-
ACTL8 Promotes the Progression of Gastric Cancer Through PI3K/AKT/mTOR Signaling Pathway.Dig Dis Sci. 2024 Oct;69(10):3786-3798. doi: 10.1007/s10620-024-08649-6. Epub 2024 Sep 25. Dig Dis Sci. 2024. PMID: 39322809 Free PMC article.
-
Cleavage stimulation factor 2 promotes malignant progression of liver hepatocellular carcinoma by activating phosphatidylinositol 3'-kinase/protein kinase B/mammalian target of rapamycin pathway.Bioengineered. 2022 Apr;13(4):10047-10060. doi: 10.1080/21655979.2022.2063100. Bioengineered. 2022. PMID: 35412944 Free PMC article.
-
PIK3CA mutation confers resistance to chemotherapy in triple-negative breast cancer by inhibiting apoptosis and activating the PI3K/AKT/mTOR signaling pathway.Ann Transl Med. 2021 Mar;9(5):410. doi: 10.21037/atm-21-698. Ann Transl Med. 2021. PMID: 33842631 Free PMC article.
-
Targeting the PI3K/AKT/mTOR pathway in triple-negative breast cancer: a review.Breast Cancer Res Treat. 2018 Jun;169(3):397-406. doi: 10.1007/s10549-018-4697-y. Epub 2018 Feb 7. Breast Cancer Res Treat. 2018. PMID: 29417298 Review.
-
Treatment strategies targeting the phosphoinositide 3-kinase/protein kinase B/mechanistic target of rapamycin pathway against triple-negative breast cancer.World J Clin Oncol. 2025 May 24;16(5):104623. doi: 10.5306/wjco.v16.i5.104623. World J Clin Oncol. 2025. PMID: 40503407 Free PMC article. Review.
Cited by
-
Circular RNA UBAP2 facilitates the cisplatin resistance of triple-negative breast cancer via microRNA-300/anti-silencing function 1B histone chaperone/PI3K/AKT/mTOR axis.Bioengineered. 2022 Mar;13(3):7197-7208. doi: 10.1080/21655979.2022.2036894. Bioengineered. 2022. PMID: 35263216 Free PMC article.
-
ACTL8 Promotes the Progression of Gastric Cancer Through PI3K/AKT/mTOR Signaling Pathway.Dig Dis Sci. 2024 Oct;69(10):3786-3798. doi: 10.1007/s10620-024-08649-6. Epub 2024 Sep 25. Dig Dis Sci. 2024. PMID: 39322809 Free PMC article.
-
Osteopontin promotes tumor growth and metastasis and GPX4-mediated anti-lipid peroxidation in triple-negative breast cancer by activating the PI3k/Akt/mTOR pathway.J Cancer Res Clin Oncol. 2024 Mar 25;150(3):155. doi: 10.1007/s00432-024-05658-w. J Cancer Res Clin Oncol. 2024. PMID: 38526702 Free PMC article.
-
Synergistic bioinformatics and sophisticated machine learning unveil ferroptosis-driven regulatory pathways and immunotherapy potential in breast carcinoma.Discov Oncol. 2025 May 4;16(1):668. doi: 10.1007/s12672-025-02393-7. Discov Oncol. 2025. PMID: 40320501 Free PMC article.
-
FAP promotes clear cell renal cell carcinoma progression via activating the PI3K/AKT/mTOR signaling pathway.Cancer Cell Int. 2023 Sep 27;23(1):217. doi: 10.1186/s12935-023-03073-8. Cancer Cell Int. 2023. PMID: 37752545 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

