Mapping the expression of transient receptor potential channels across murine placental development
- PMID: 33884443
- PMCID: PMC8233283
- DOI: 10.1007/s00018-021-03837-3
Mapping the expression of transient receptor potential channels across murine placental development
Abstract
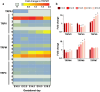
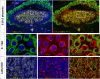
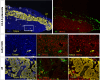
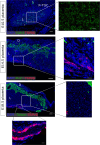
Transient receptor potential (TRP) channels play prominent roles in ion homeostasis by their ability to control cation influx. Mouse placentation is governed by the processes of trophoblast proliferation, invasion, differentiation, and fusion, all of which require calcium signaling. Although certain TRP channels have been shown to contribute to maternal-fetal transport of magnesium and calcium, a role for TRP channels in specific trophoblast functions has been disregarded. Using qRT-PCR and in situ hybridisation, the spatio-temporal expression pattern of TRP channels in the mouse placenta across gestation (E10.5-E18.5) was assessed. Prominent expression was observed for Trpv2, Trpm6, and Trpm7. Calcium microfluorimetry in primary trophoblast cells isolated at E14.5 of gestation further revealed the functional activity of TRPV2 and TRPM7. Finally, comparing TRP channels expression in mouse trophoblast stem cells (mTSCs) and mouse embryonic stem cells (mESC) confirmed the specific expression of TRPV2 during placental development. Moreover, TRP channel expression was similar in mTSCs compared to primary trophoblasts and validate mTSC as a model to study TRP channels in placental development. Collectivity, our results identify a specific spatio-temporal TRP channel expression pattern in trophoblasts, suggesting a possible involvement in regulating the process of placentation.
Keywords: Placental development; Primary trophoblast cells; TRP channels; Trophoblast stem cells.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
Similar articles
-
Tissue-specific expression of TRP channel genes in the mouse and its variation in three different mouse strains.BMC Genomics. 2006 Jun 20;7:159. doi: 10.1186/1471-2164-7-159. BMC Genomics. 2006. PMID: 16787531 Free PMC article.
-
HGF/c-Met signaling regulates early differentiation of placental trophoblast cells.J Reprod Dev. 2021 Apr 21;67(2):89-97. doi: 10.1262/jrd.2020-107. Epub 2021 Jan 15. J Reprod Dev. 2021. PMID: 33455972 Free PMC article.
-
Functional characterization of transient receptor potential channels in mouse urothelial cells.Am J Physiol Renal Physiol. 2010 Mar;298(3):F692-701. doi: 10.1152/ajprenal.00599.2009. Epub 2009 Dec 16. Am J Physiol Renal Physiol. 2010. PMID: 20015940 Free PMC article.
-
Progress in deciphering trophoblast cell differentiation during human placentation.Curr Opin Cell Biol. 2020 Dec;67:86-91. doi: 10.1016/j.ceb.2020.08.010. Epub 2020 Sep 18. Curr Opin Cell Biol. 2020. PMID: 32957014 Review.
-
Establishing life is a calcium-dependent TRiP: Transient receptor potential channels in reproduction.Biochim Biophys Acta Mol Cell Res. 2018 Nov;1865(11 Pt B):1815-1829. doi: 10.1016/j.bbamcr.2018.08.005. Epub 2018 Aug 9. Biochim Biophys Acta Mol Cell Res. 2018. PMID: 30798946 Review.
Cited by
-
TRPM4 and TRPV2 are two novel prognostic biomarkers and promising targeted therapy in UVM.Front Mol Biosci. 2022 Aug 23;9:985434. doi: 10.3389/fmolb.2022.985434. eCollection 2022. Front Mol Biosci. 2022. PMID: 36081847 Free PMC article.
-
Menthol, a consumer product additive, adversely affects human embryonic stem cells via activation of TRPM8 and TRPA1 channels.Stem Cells Transl Med. 2025 Mar 18;14(3):szae099. doi: 10.1093/stcltm/szae099. Stem Cells Transl Med. 2025. PMID: 40139559 Free PMC article.
-
TRP channel expression correlates with the epithelial-mesenchymal transition and high-risk endometrial carcinoma.Cell Mol Life Sci. 2021 Dec 22;79(1):26. doi: 10.1007/s00018-021-04023-1. Cell Mol Life Sci. 2021. PMID: 34936030 Free PMC article.
-
Transient Receptor Potential Channels in the Epithelial-to-Mesenchymal Transition.Int J Mol Sci. 2021 Jul 30;22(15):8188. doi: 10.3390/ijms22158188. Int J Mol Sci. 2021. PMID: 34360952 Free PMC article. Review.
-
New Insights into TRP Ion Channels in Stem Cells.Int J Mol Sci. 2022 Jul 14;23(14):7766. doi: 10.3390/ijms23147766. Int J Mol Sci. 2022. PMID: 35887116 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

