Influence of atorvastatin on metabolic pattern of rats with pulmonary hypertension
- PMID: 33886502
- PMCID: PMC8109122
- DOI: 10.18632/aging.202898
Influence of atorvastatin on metabolic pattern of rats with pulmonary hypertension
Abstract
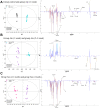
Background: Metabonomics has been widely used to analyze the initiation, progress, and development of diseases. However, application of metabonomics to explore the mechanism of pulmonary arterial hypertension (PAH) are poorly reported. This study aimed to investigate the influence of atorvastatin (Ato) on metabolic pattern of rats with pulmonary hypertension.
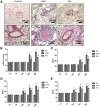
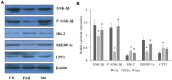
Methods: PAH animal model was established using monocrotaline (MCT). The mean pulmonary artery pressure (mPAP) and right ventricular hypertrophy index (RVHI) were measured. The microstructure of pulmonary arterioles was observed by HE staining. Nuclear magnetic resonance was used to detect and analyze the serum metabolites. The levels of glycogen synthase kinase-3β (GSK-3β), hexokinase 2 (HK-2), sterol regulatory element-binding protein 1c (SREBP-1c), and carnitine palmitoyltransferase I (CPT-1) in the lung tissues were measured.
Results: Ato significantly improved lung function by decreasing mPAP, RVHI, wall thickness, and wall area. Differences in metabolic patterns were observed among normal, PAH, and Ato group. The levels of GSK-3β and SREBP-1c were decreased, but HK-2 and CPT-1 were increased in the group PAH. Ato treatment markedly reversed the influence of MCT.
Conclusion: Ato significantly improved the pulmonary vascular remodeling and pulmonary hypertension of PAH rats due to its inhibition on Warburg effect and fatty acid β oxidation.
Keywords: Warburg effect; fatty acid β oxidation; metabonomics; monocrotaline; pulmonary arterial hypertension.
Conflict of interest statement
Figures




Similar articles
-
Preventive and remedial application of etanercept attenuate monocrotaline-induced pulmonary arterial hypertension.Int J Rheum Dis. 2016 Feb;19(2):192-8. doi: 10.1111/1756-185X.12304. Epub 2014 Feb 25. Int J Rheum Dis. 2016. PMID: 24612527
-
Activation of AMPK prevents monocrotaline-induced pulmonary arterial hypertension by suppression of NF-κB-mediated autophagy activation.Life Sci. 2018 Sep 1;208:87-95. doi: 10.1016/j.lfs.2018.07.018. Epub 2018 Jul 17. Life Sci. 2018. PMID: 30009823
-
Effects of atorvastatin and losartan on monocrotaline-induced pulmonary artery remodeling in rats.Clin Exp Hypertens. 2010;32(8):547-54. doi: 10.3109/10641963.2010.503295. Clin Exp Hypertens. 2010. PMID: 21091363
-
Tetrandrine prevents monocrotaline-induced pulmonary arterial hypertension in rats through regulation of the protein expression of inducible nitric oxide synthase and cyclic guanosine monophosphate-dependent protein kinase type 1.J Vasc Surg. 2016 Nov;64(5):1468-1477. doi: 10.1016/j.jvs.2015.09.016. J Vasc Surg. 2016. PMID: 26527422
-
Metabolism in Pulmonary Hypertension.Annu Rev Physiol. 2021 Feb 10;83:551-576. doi: 10.1146/annurev-physiol-031620-123956. Annu Rev Physiol. 2021. PMID: 33566674 Free PMC article. Review.
Cited by
-
Pharmacometabolomics for the Study of Lipid-Lowering Therapies: Opportunities and Challenges.Int J Mol Sci. 2023 Feb 7;24(4):3291. doi: 10.3390/ijms24043291. Int J Mol Sci. 2023. PMID: 36834701 Free PMC article. Review.
-
The zinc transporter ZIP12 regulates monocrotaline-induced proliferation and migration of pulmonary arterial smooth muscle cells via the AKT/ERK signaling pathways.BMC Pulm Med. 2022 Mar 28;22(1):111. doi: 10.1186/s12890-022-01905-3. BMC Pulm Med. 2022. PMID: 35346134 Free PMC article.
-
Metabolomics in Pulmonary Hypertension-A Useful Tool to Provide Insights into the Dark Side of a Tricky Pathology.Int J Mol Sci. 2023 Aug 25;24(17):13227. doi: 10.3390/ijms241713227. Int J Mol Sci. 2023. PMID: 37686034 Free PMC article.
-
The contributory role of GSK3β in hypertension exacerbating atherosclerosis by regulating the OMA1/PGC1α pathway.Apoptosis. 2025 Feb;30(1-2):117-130. doi: 10.1007/s10495-024-02029-1. Epub 2024 Oct 19. Apoptosis. 2025. PMID: 39427090
-
Hypoxia-induced histone lactylation promotes pulmonary arterial smooth muscle cells proliferation in pulmonary hypertension.Mol Cell Biochem. 2025 Jun 30. doi: 10.1007/s11010-025-05342-8. Online ahead of print. Mol Cell Biochem. 2025. PMID: 40588665
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

